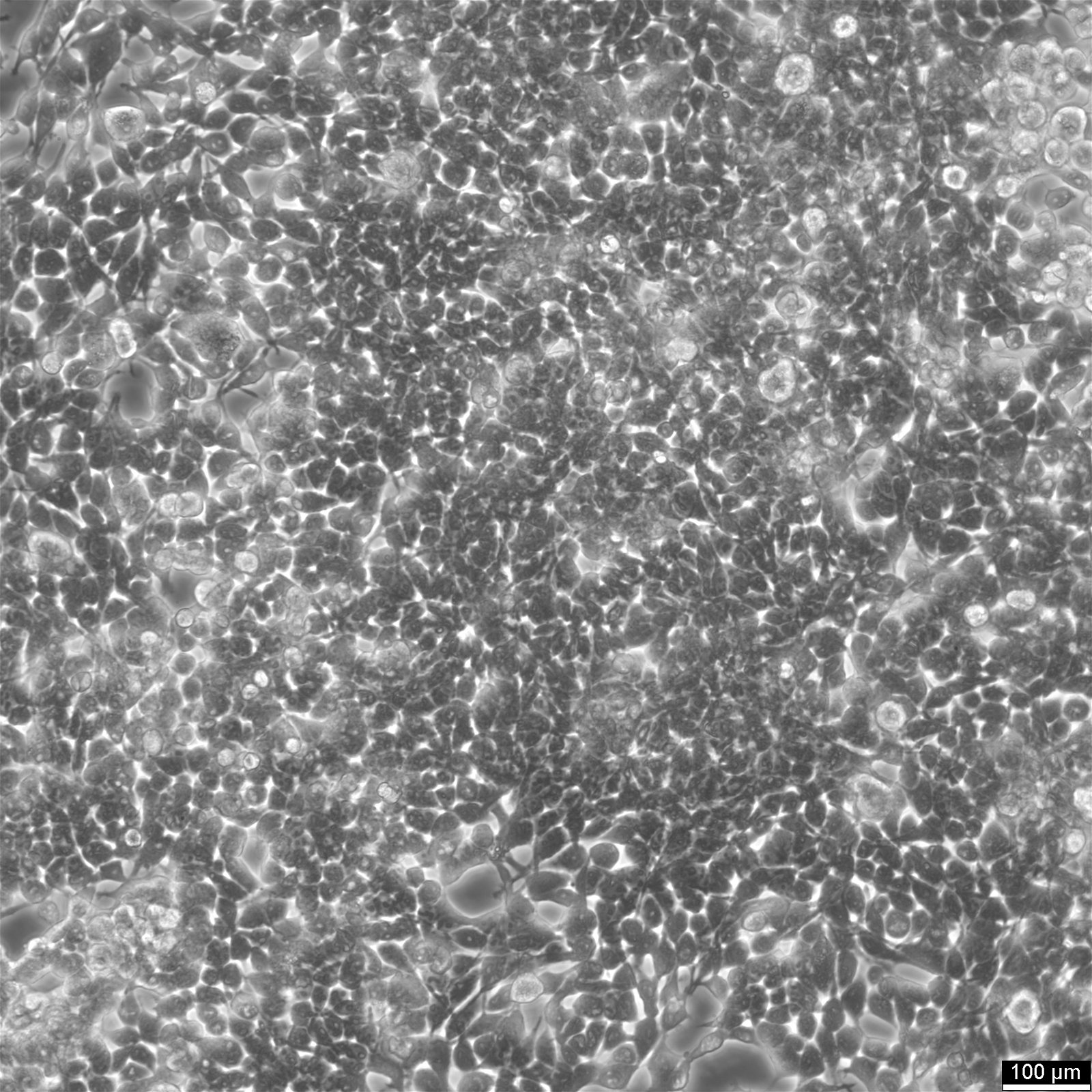
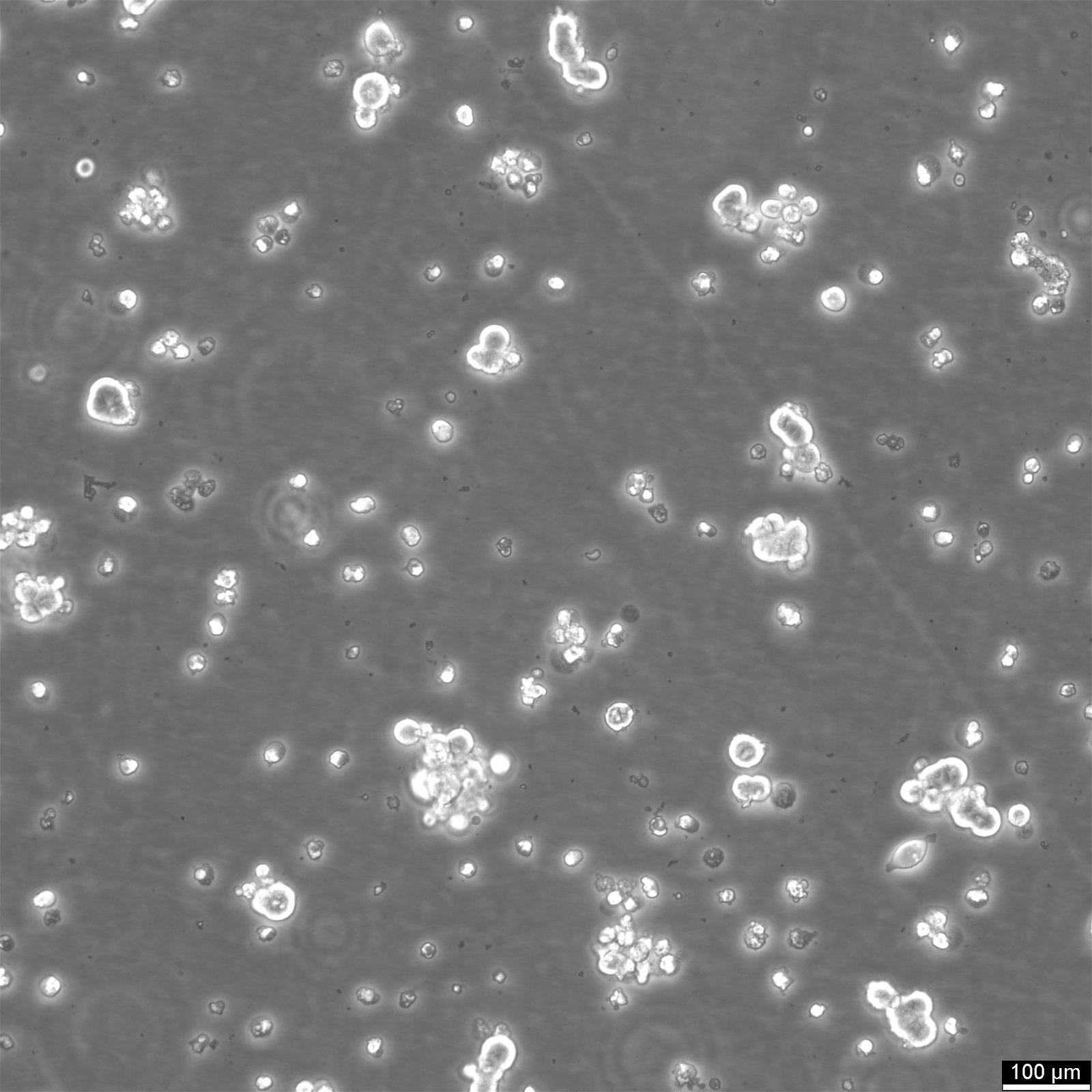
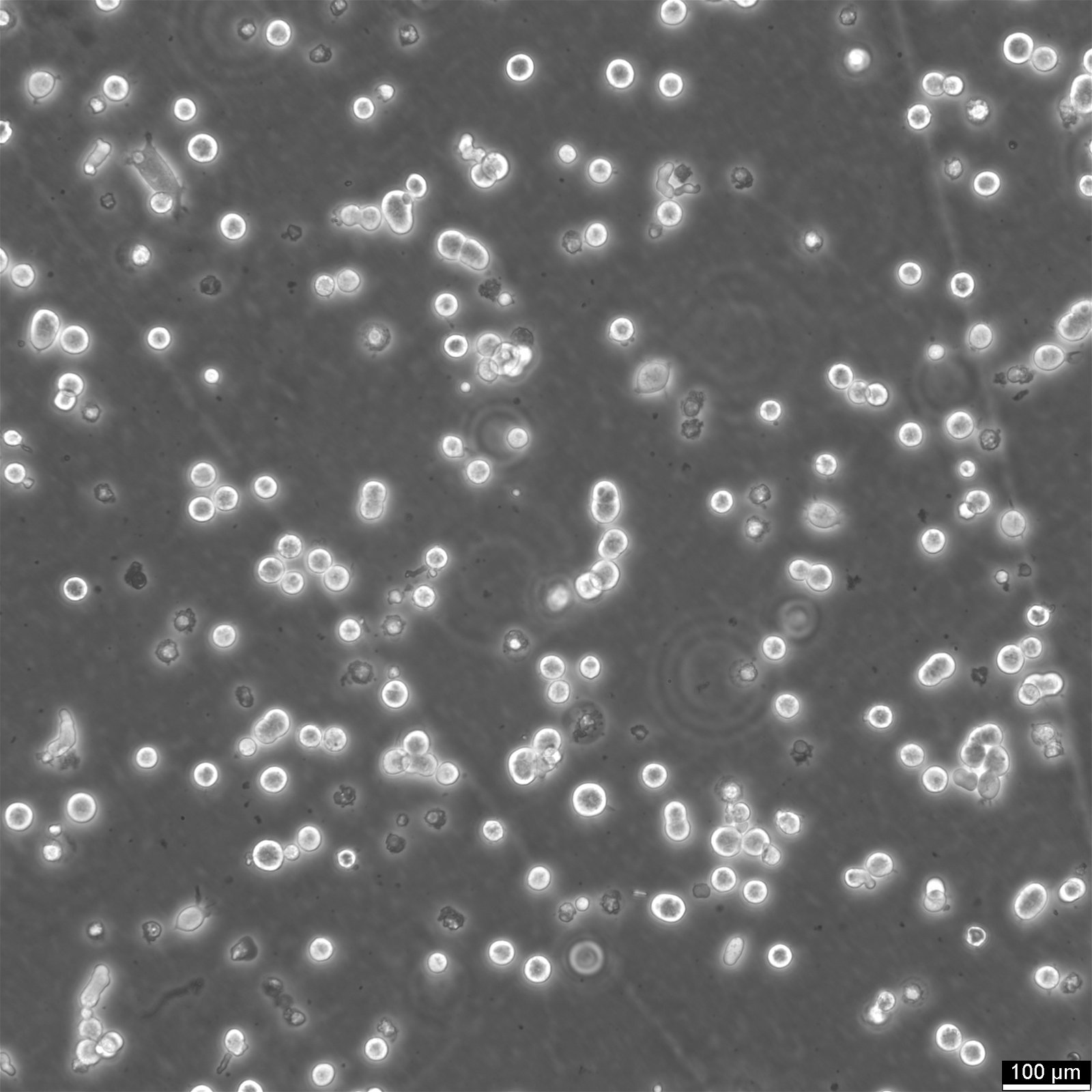
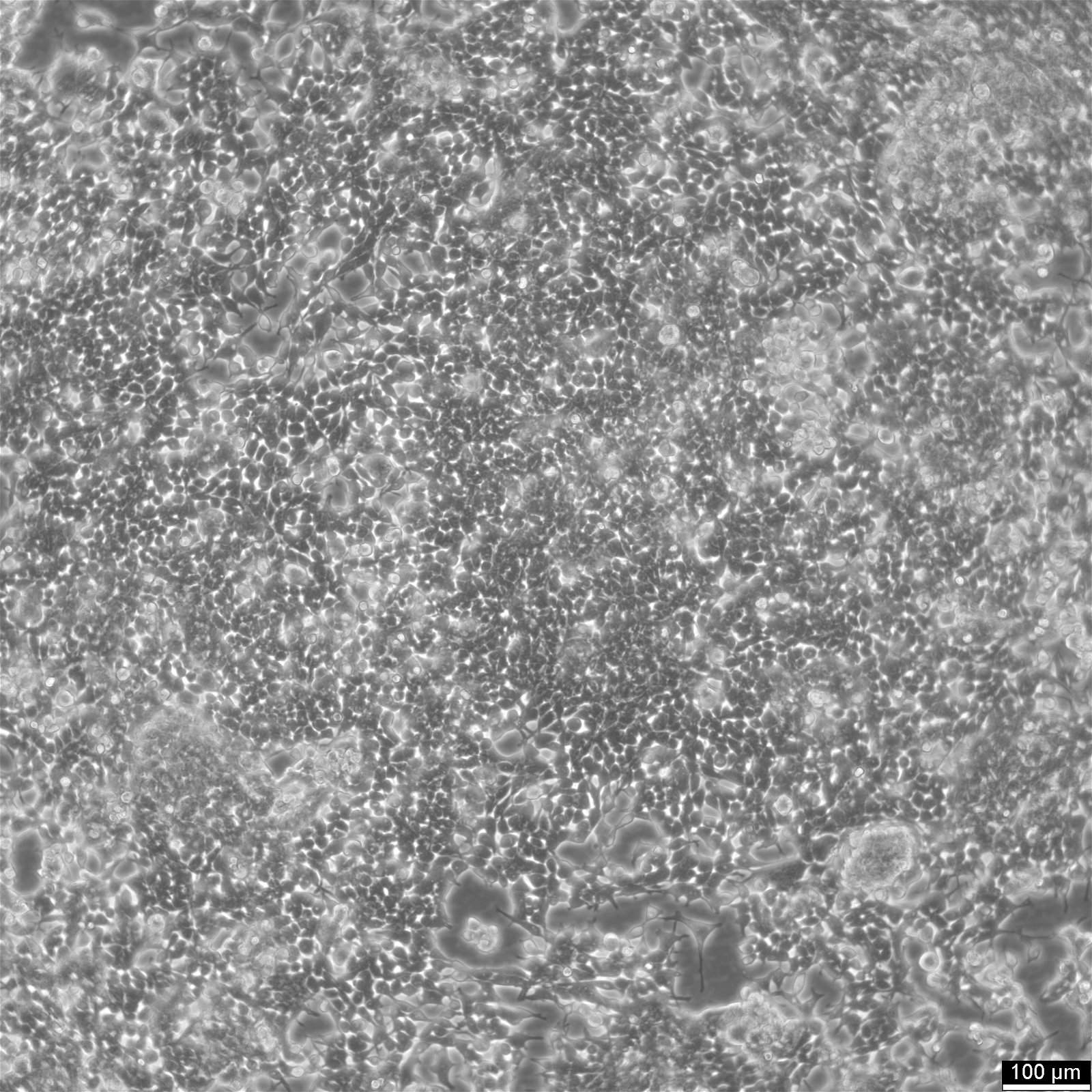
HEK293 EBNA Cells




















Product number:
300264
General information
| Description | Large-scale transfection of mammalian cells has emerged as an indispensable technology within the scientific community. It allows for the efficient production of milligram-to-gram quantities of r-proteins within a short timeframe, just a few days after the cloning of cDNAs into the suitable expression vector. Among the various cell lines available, the HEK293 cell line stably expressing the Epstein-Barr virus nuclear antigen-1 (HEK293-EBNA1, or 293E) stands out as the most widely utilized cell line for large-scale transfection experiments. One of the critical advantages of utilizing HEK293-EBNA1 cells is their compatibility with expression vectors carrying the Epstein-Barr virus origin of replication, oriP, such as the pTT vector. This compatibility leads to a significant threefold improvement in the yield of r-proteins compared to using a non-oriP vector. Leveraging this potential, researchers can now achieve higher yields of their desired r-proteins, bringing them closer to their research and development goals. |
|---|---|
| Organism | Human |
| Tissue | Embryonic kidney |
| Synonyms | HEK293-EBNA, 293 c18, 293c18, HEK 293 c18, HEK-293 c18, HEK293-EBNA1, HEK-293-EBNA, HEK 293-EBNA, HEK 293 EBNA, HEK293EBNA, 293 EBNA, 293-EBNA1, 293-EBNA, 293/EBNA, 293EBNA, EBNA-293, EBNA293, HEK293E, HEK/EBNA, HEK-EBNA, HEK.EBNA, 293/EBNA-1 |
Characteristics
| Age | Fetus |
|---|---|
| Gender | Female |
| Morphology | Epithelial |
| Growth properties | Adherent |
Identifiers / Biosafety / Citation
| Citation | HEK293 EBNA (Cytion catalog number 300264) |
|---|---|
| Biosafety level | 2 |
Expression / Mutation
| Antigen expression | EBNA1 |
|---|---|
| Viruses | Adenovirus 5 (Transformant), EBV (expresses EBNA1) |
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | HEK293 EBNA cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
