CAL 27 Cells
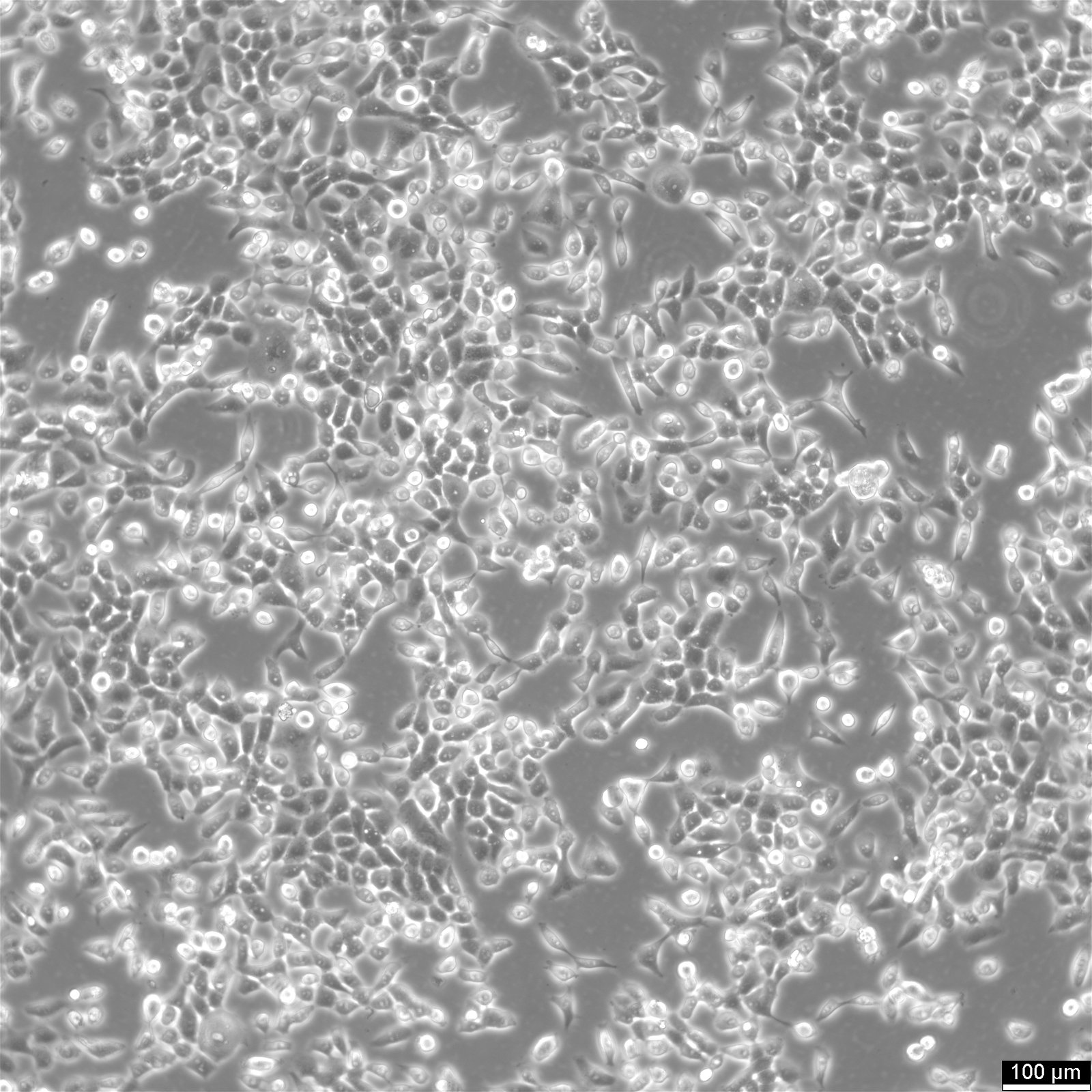
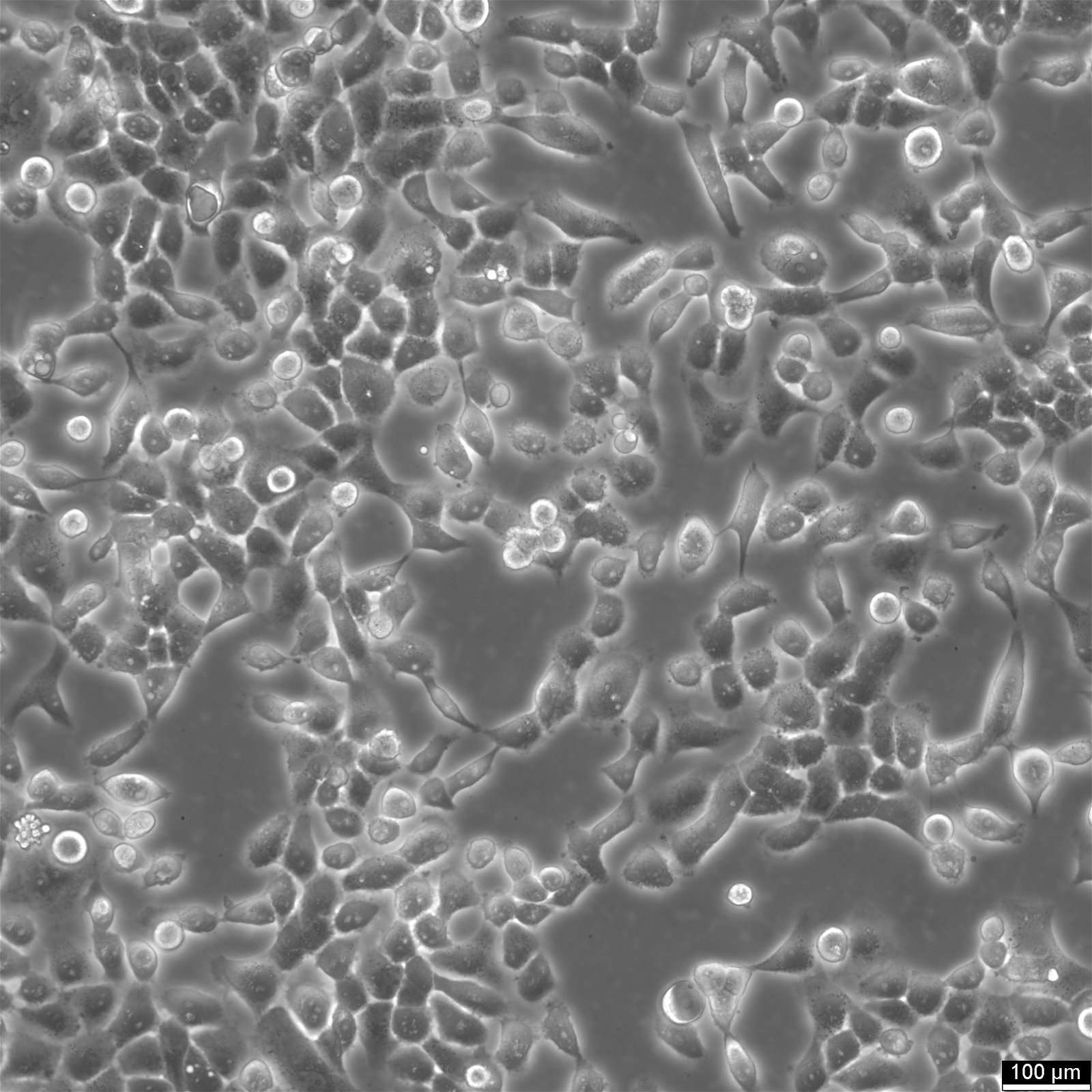






Basic details about Cal 27 cells
| Description | Cal 27 cells is a human squamous cell carcinoma cell line derived from a primary tumor located in the tongue of a 56-year-old male in 1982. Cal 27 cells are epithelial in morphology and are widely used in scientific research to study oral carcinogenesis, the biology of squamous cell and oropharyngeal carcinoma, and to evaluate potential therapeutic agents for head and neck cancers. The Cal27 cell line has been employed in a variety of research applications, including studies on cell proliferation, apoptosis, particularly in the context of anticancer drug sensitivity and the search for novel anticancer agents, migration, and invasion. They have also been used to investigate the effects of various chemotherapeutic agents such as Cisplatin, radiation therapy, and targeted therapies. The Cal-27 adenosquamous carcinoma cell line is further used as xenografts, which are instrumental for studying tumor angiogenesis, lymph node metastasis, as well as metastasis and chemoresistance mechanisms. The interaction of Cal27 cells with integrins α6β4 and αvβ3 is of interest, as these molecules play a crucial role in cell adhesion. Studies have explored the effects of targeting these pathways with drugs like vismodegib and itraconazole, substances known to modulate the hedgehog pathway. Overall, the Cal 27 cell line serves as a robust model for investigating the complex biology of oral squamous cell carcinomas and for testing new therapeutic interventions, thereby contributing to advancements in the management and treatment of oral cancers. |
|---|---|
| Organism | Human |
| Tissue | Tongue |
| Disease | Tongue squamous cell carcinoma |
| Synonyms | Cal-27, CAL 27, Cal 27, CAL27, Cal27, Centre Antoine Lacassagne-27 |
Features of the CAL-27 carcinoma cell line
| Age | 56 years |
|---|---|
| Gender | Male |
| Morphology | Epithelial |
| Growth properties | Adherent |
Specification
| Citation | CAL 27 (Cytion catalog number 305029) |
|---|---|
| Biosafety level | 1 |
Genotype
| Tumorigenic | Yes |
|---|
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:2 to 1:4 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | CAL 27 cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
CAL 27 cell purity and identity checks
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|---|
| STR profile |
Amelogenin: x,x
CSF1PO: 10
D13S317: 10
D16S539: 11
D5S818: 11
D7S820: 10
TH01: 6
TPOX: 8
vWA: 14
D3S1358: 16
D21S11: 28
D18S51: 13
Penta E: 7
Penta D: 9
D8S1179: 13
FGA: 25
|
