BHT101 Cells
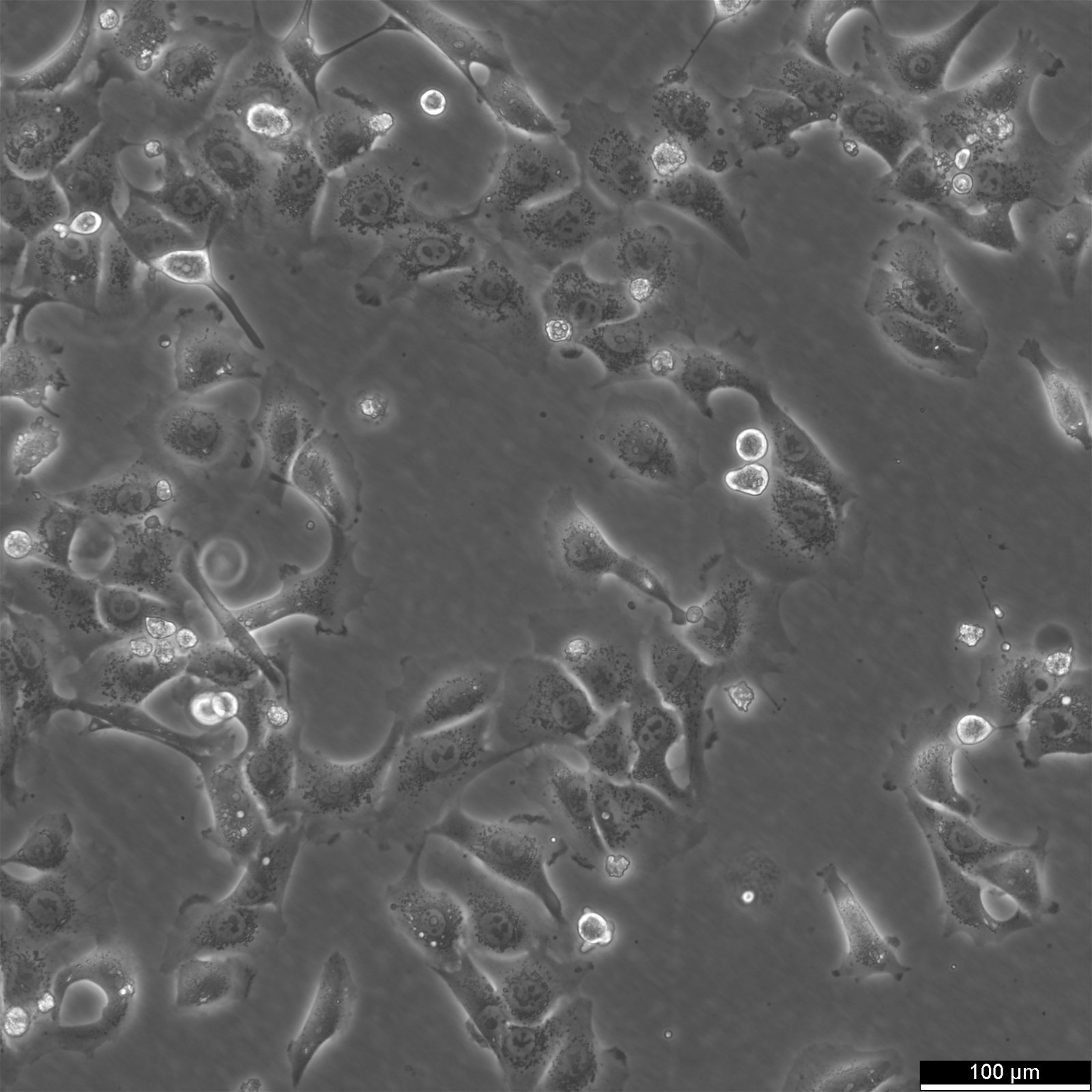
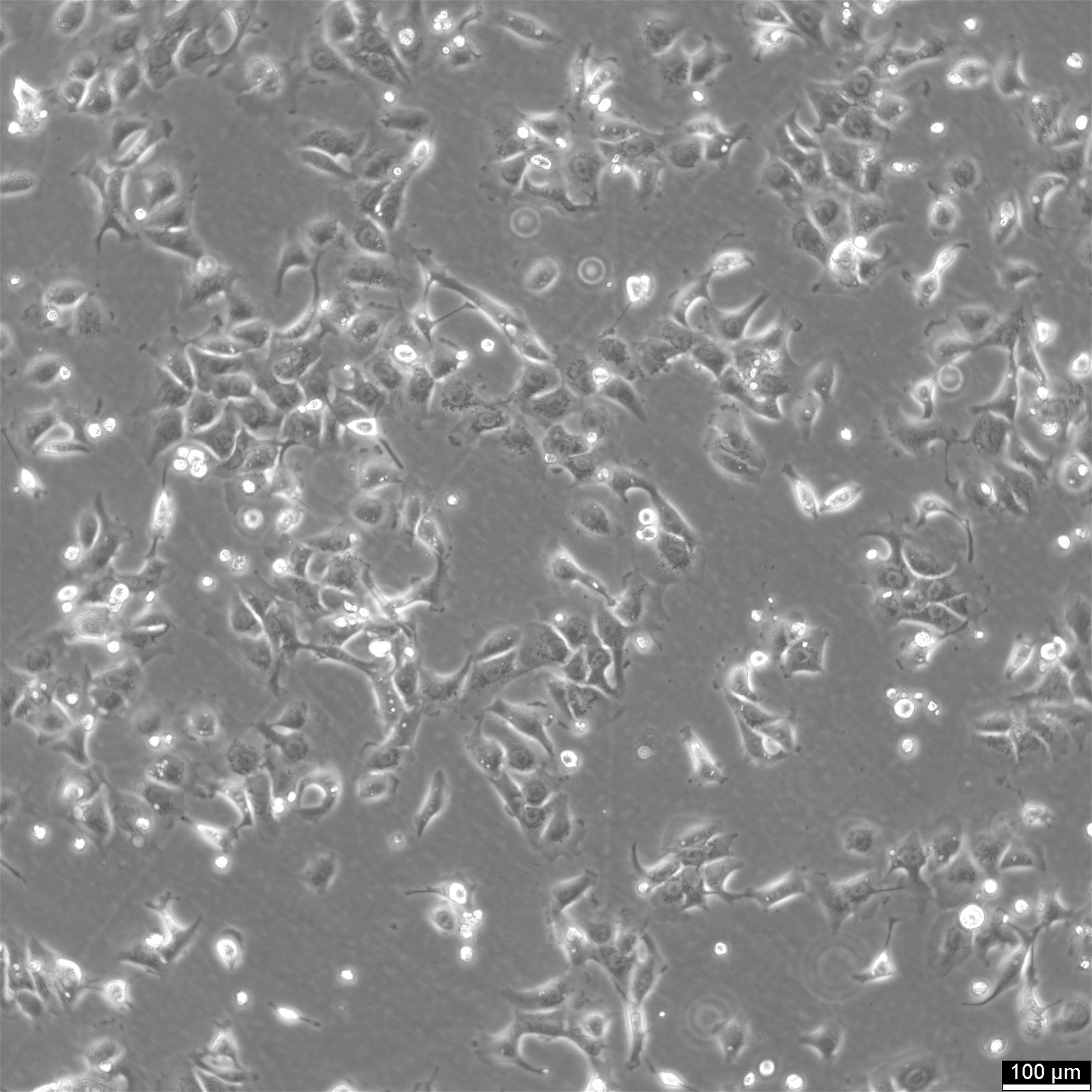
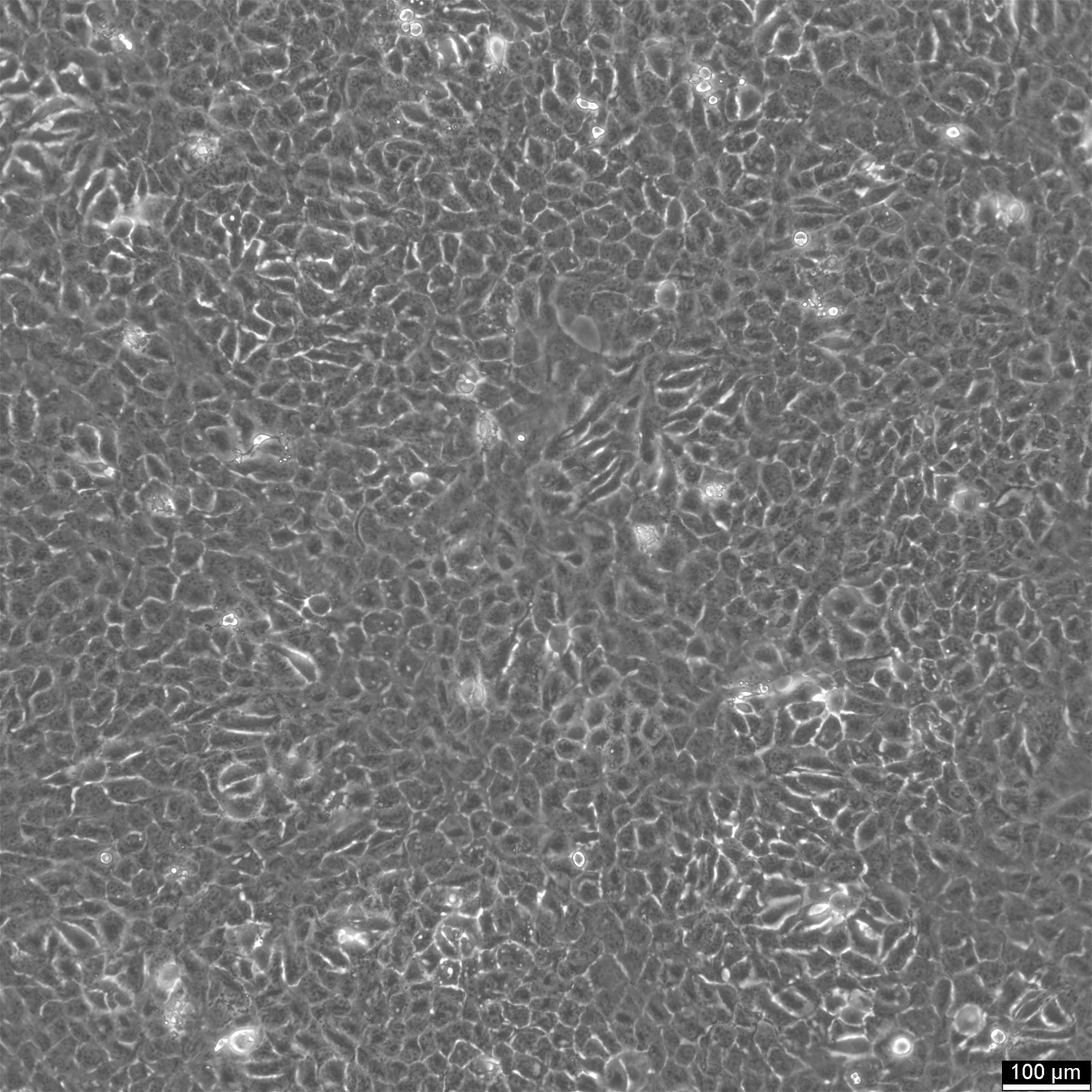
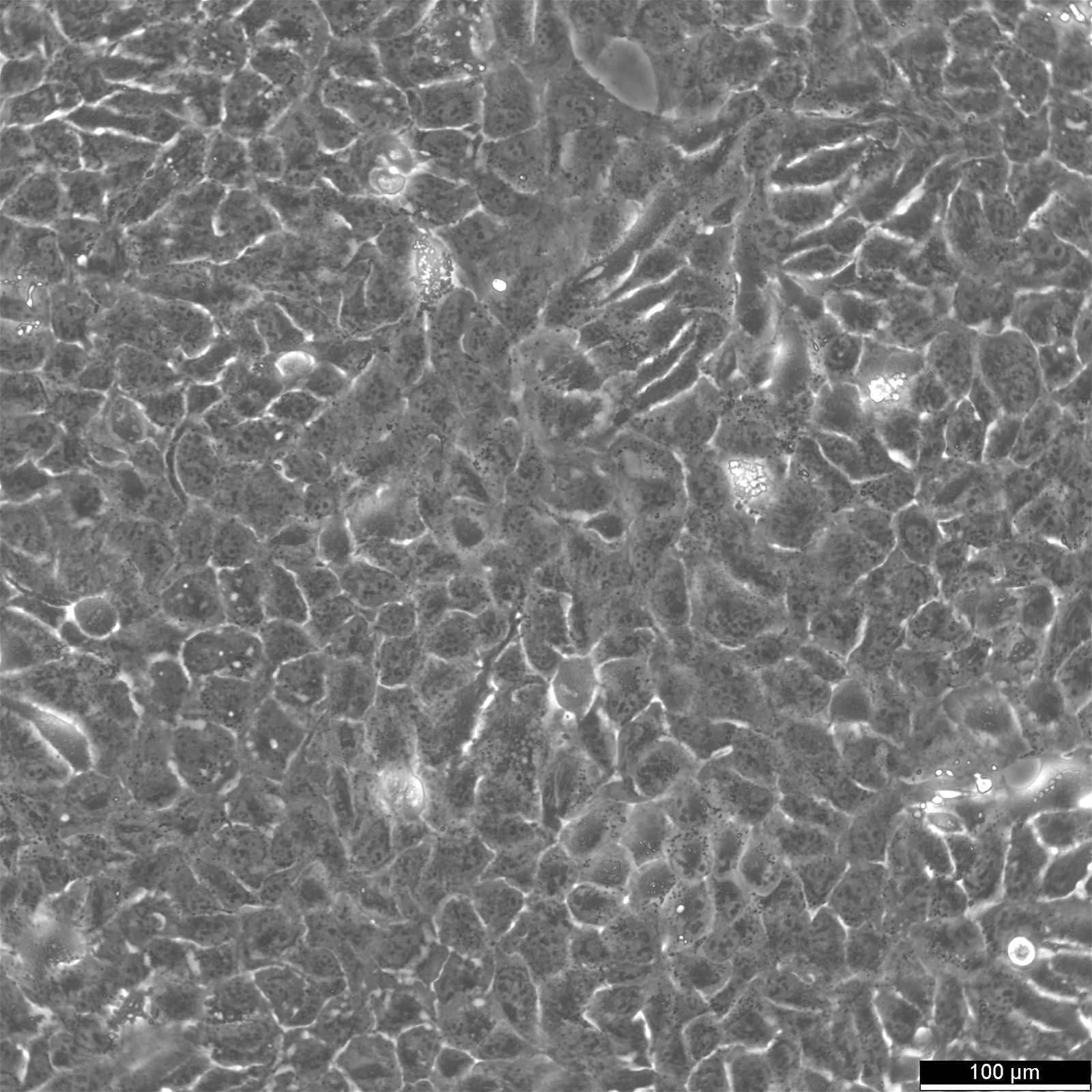












Product number:
305112
General information
| Description | This cell line was established from the lymph node metastasis of a 63-year-old woman with anaplastic papillary thyroid carinoma. Cells were described in the literature to not produce hormones, but to be partly positive for thyroglobulin- and thyroxine (T4). |
|---|---|
| Organism | Human |
| Tissue | Thyroid |
| Disease | Anaplastic thyroid carcinoma |
| Metastatic site | Lymph node |
| Synonyms | BHT-101 |
Characteristics
| Age | 63 years |
|---|---|
| Gender | Female |
| Ethnicity | European |
| Morphology | Epithelial |
| Growth properties | Adherent |
Identifiers / Biosafety / Citation
| Citation | BHT101 (Cytion catalog number 305112) |
|---|---|
| Biosafety level | 1 |
Expression / Mutation
Handling
| Culture Medium | MEM (We do not supply this product; please consider other suppliers. Please let us know if you need further assistance) |
|---|---|
| Medium supplements | Supplement the medium with 20% heat-inactivated FBS, 5 microgram/ml human insulin, 0.005 IU/ml TSH (from Scrippslabs) - Add the required TSH just before use and sterile filter into the medium |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:2 to 1:5 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | BHT101 cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
