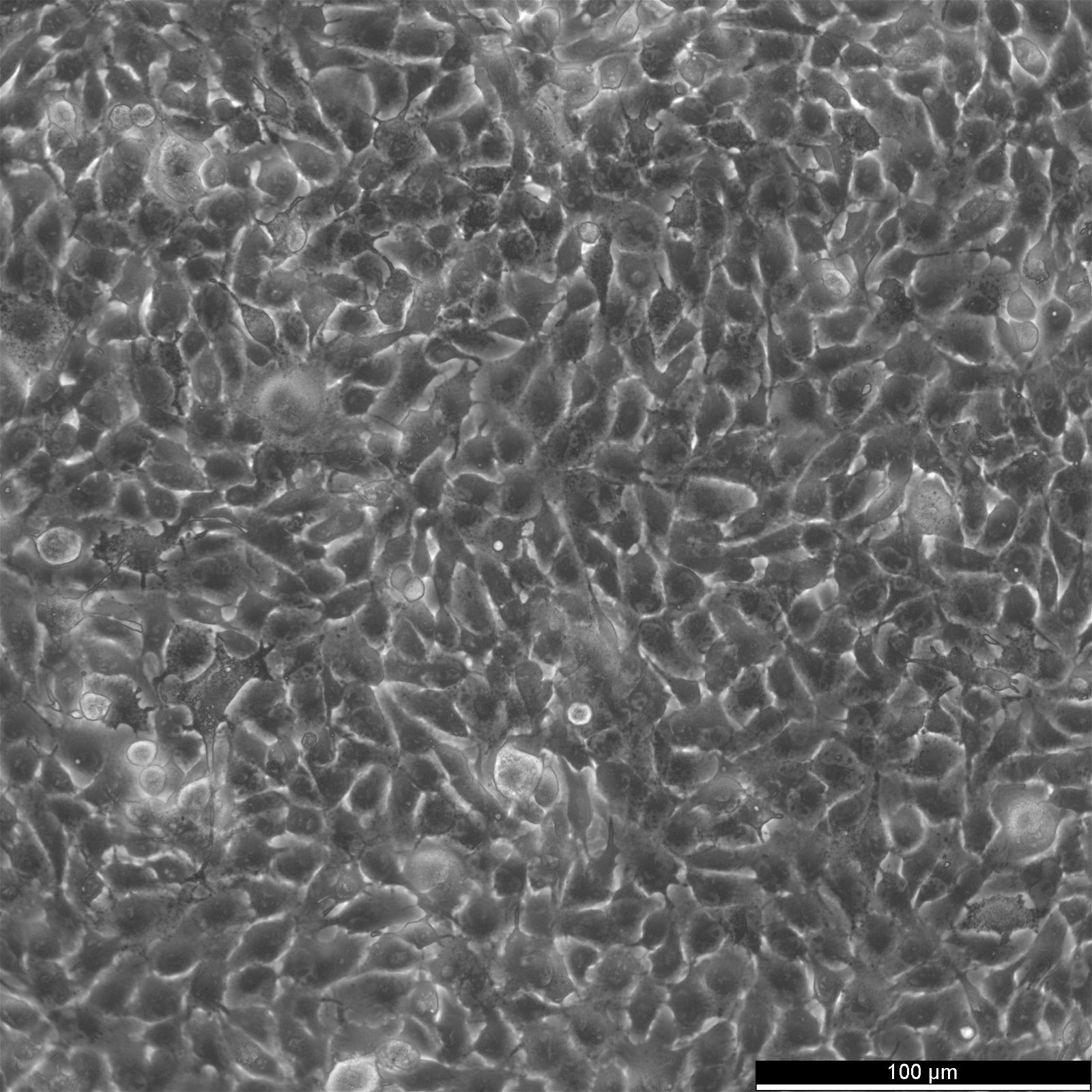
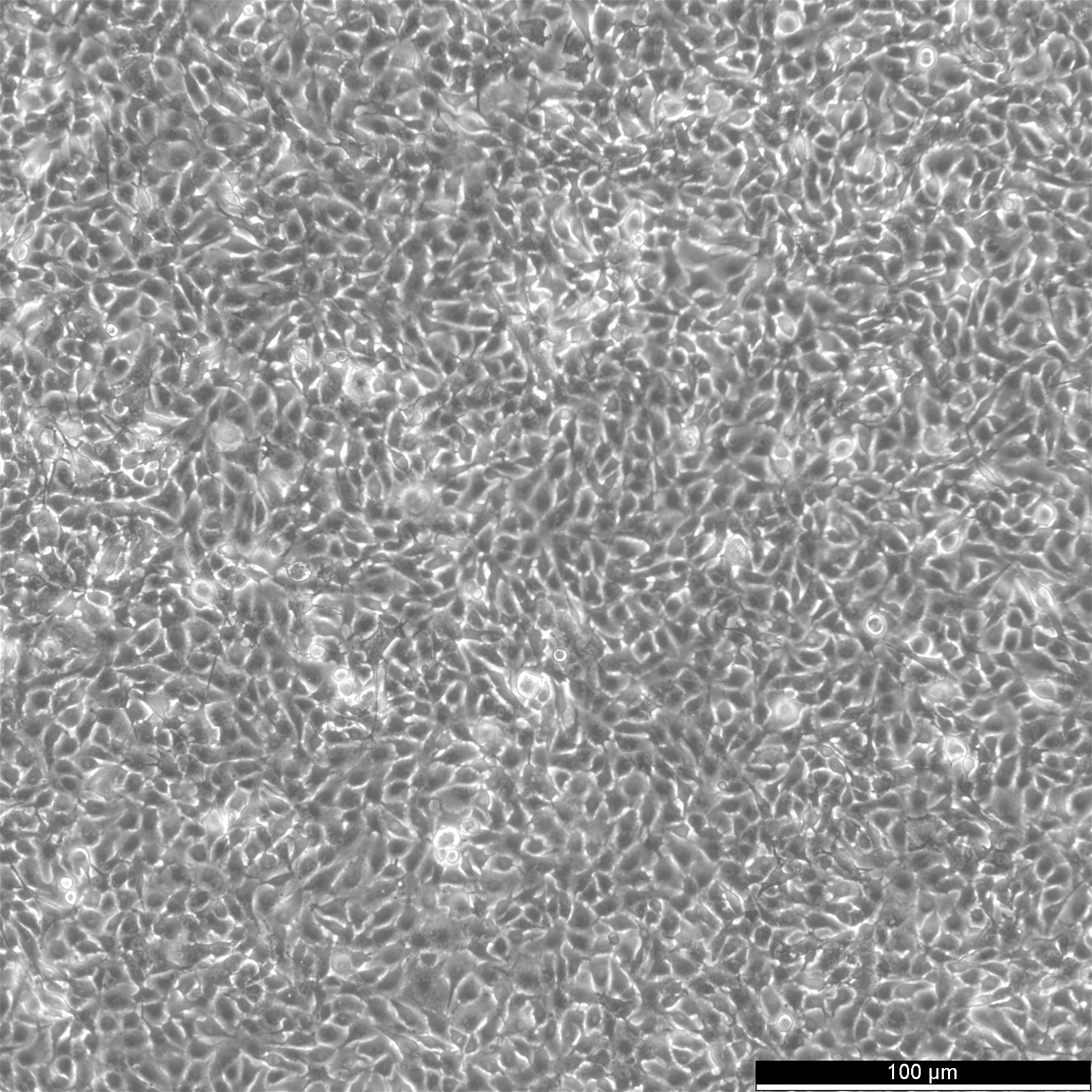
B16-F10 Cells










Product number:
305157
Key points about B16-F10 cells
| Description | The murine melanoma cell line B16-F10 is a subline of the B16 tumour line that was derived from the skin tissue of a C57BL/6J mouse. These cells exhibit a spindle-shaped and epithelial-like morphology and are often used in skin cancer research. Several sublines, for instance, B16-F1 and B16-F10, were derived from the mother B16 line by selection for their ability to form lung colonies in vivo after intravenous injection and subsequently established in vitro after one (B16-F1) or 10 (B16-F10) cycles of lung colony formation. The cell size at rest is approximately 15.4 μm with no statistical difference between B16-F1 and B16-F10 cells. The doubling times of B16-F10 cells are approximately 20.1 hours. B16-F10 possesses high lung metastatic ability, whereas B16-F1 is a subline with low metastatic potential. B16-F10 melanoma cells have been widely used as a poorly immunogenic and highly aggressive model for murine tumor immunotherapy studies. Invasive analysis shows that B16-4A5 is the most aggressive melanoma cell line for C57BL/6J's skin, followed by B16-F10 and a diminished aggressive growth pattern by the B16-GMCSF and B16-FLT3 cell lines. Furthermore, melanin is released excessively by the tumor cells, and many metastases in lung and liver parenchyma were found in studies. Regarding protein expression, inoculation of B16-F10 in mouse skin showed a perivascular predominance of VEGF expression on many vessels. VEGF is a signal protein stimulating the growth of new blood vessels. In B16-F10 inoculated test groups, S100, a protein associated with melanomas, was found heterogeneous with moderate to intense positivity. |
|---|---|
| Organism | Mouse |
| Tissue | Skin |
| Synonyms | B16/F10, B16 F10, B16F10, B16 melanoma F10 |
Features of the B16-F10 cell line
| Gender | Male |
|---|---|
| Morphology | Mixture of spindle-shaped and epithelial-like cells |
| Growth properties | Adherent |
Specifications
| Citation | B16-F10 (Cytion catalog number 305157) |
|---|---|
| Biosafety level | 1 |
Genetic profile of the B16 melanoma model
Culturing methods
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:2 to 1:4 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | B16-F10 cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality verification
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
