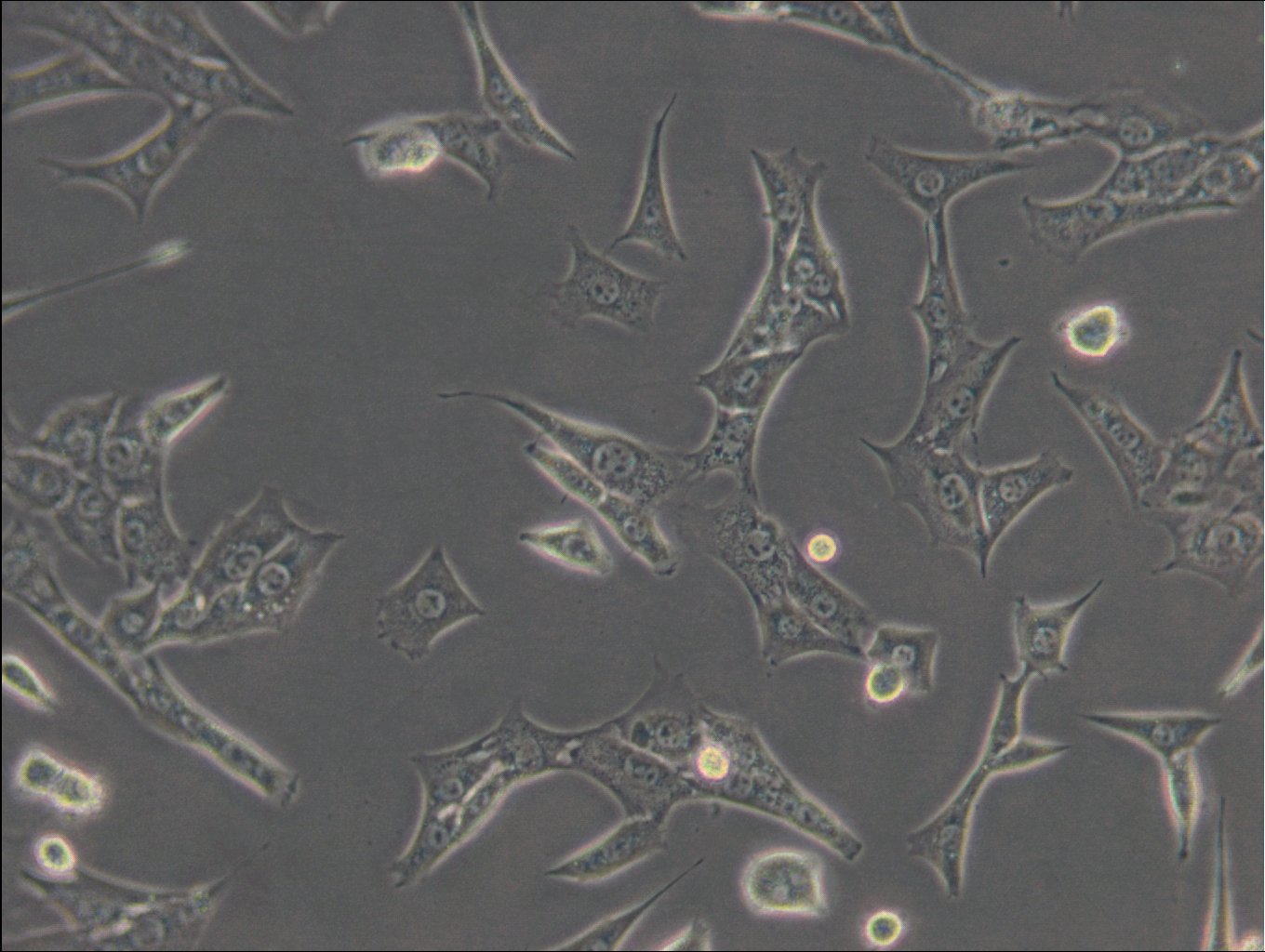
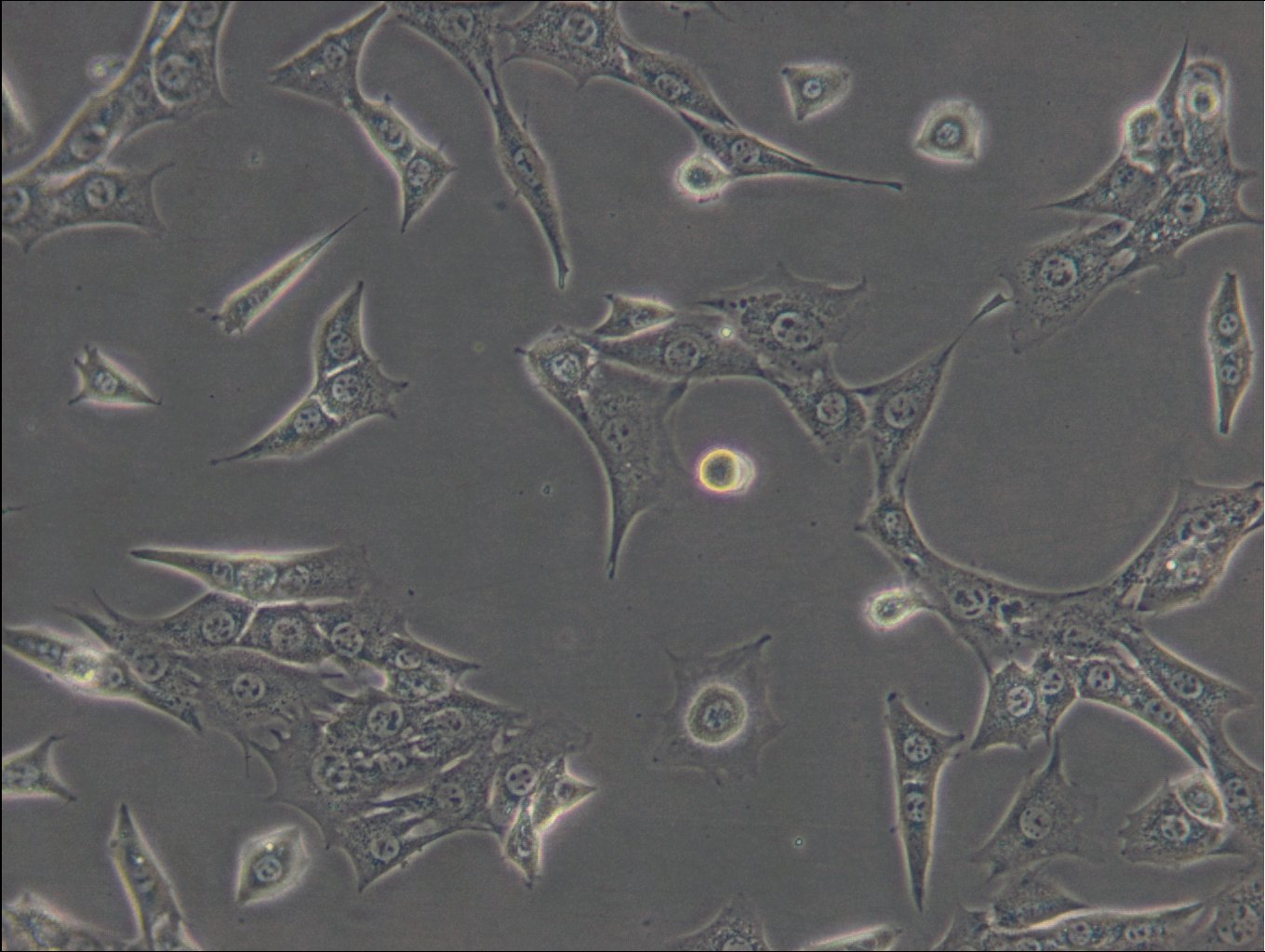
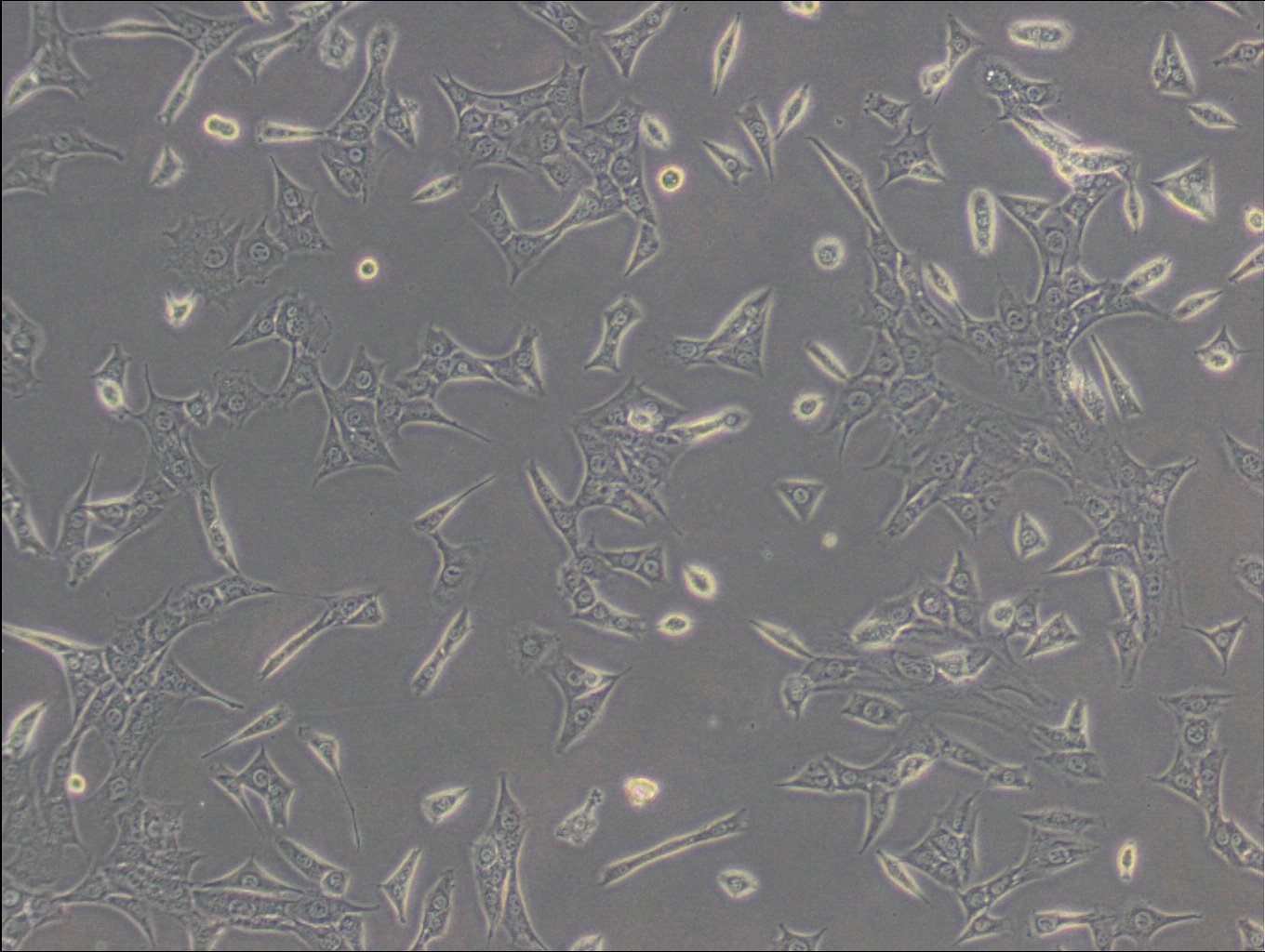
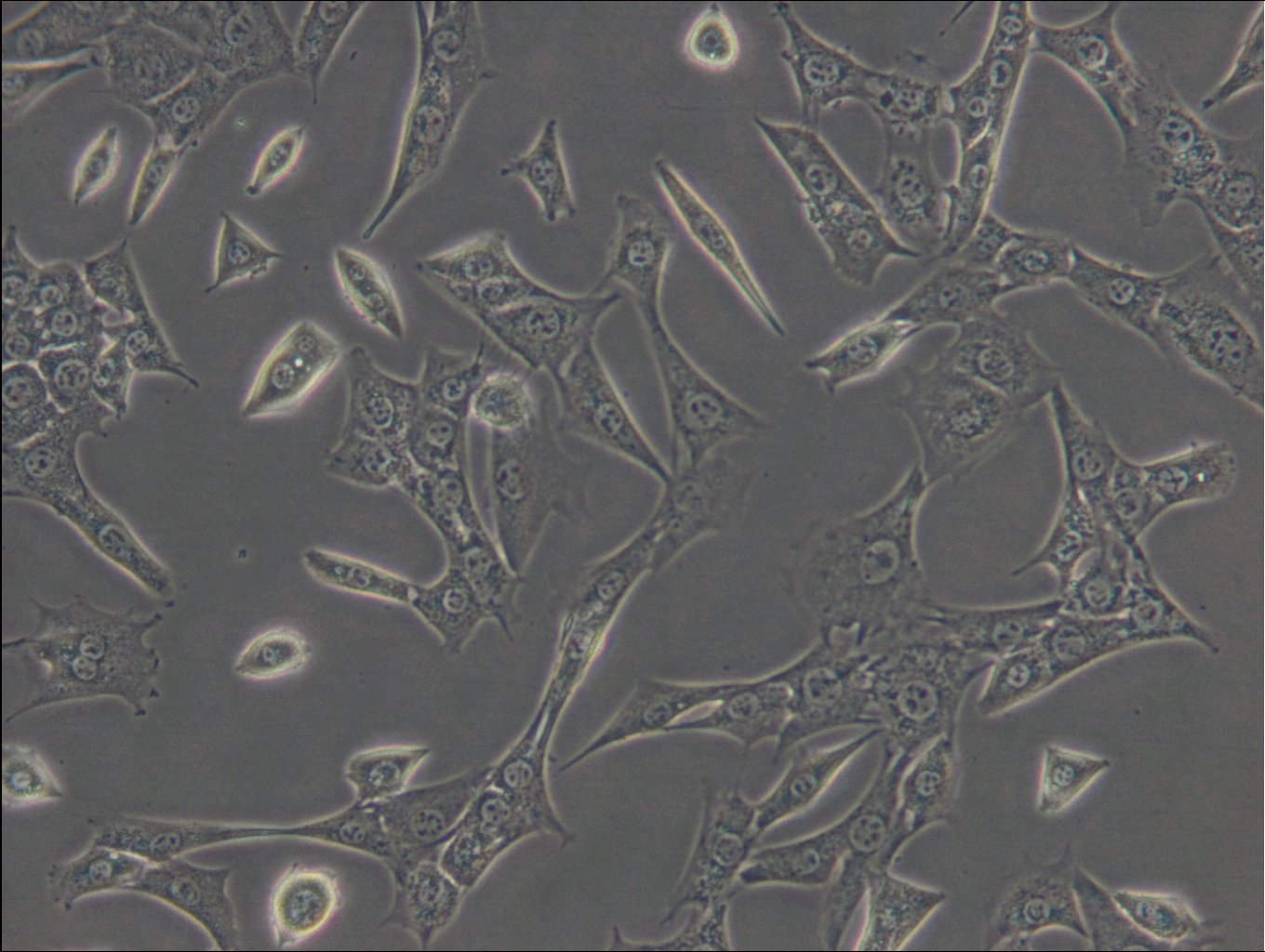
C127 Cells


















Product number:
305169
General information
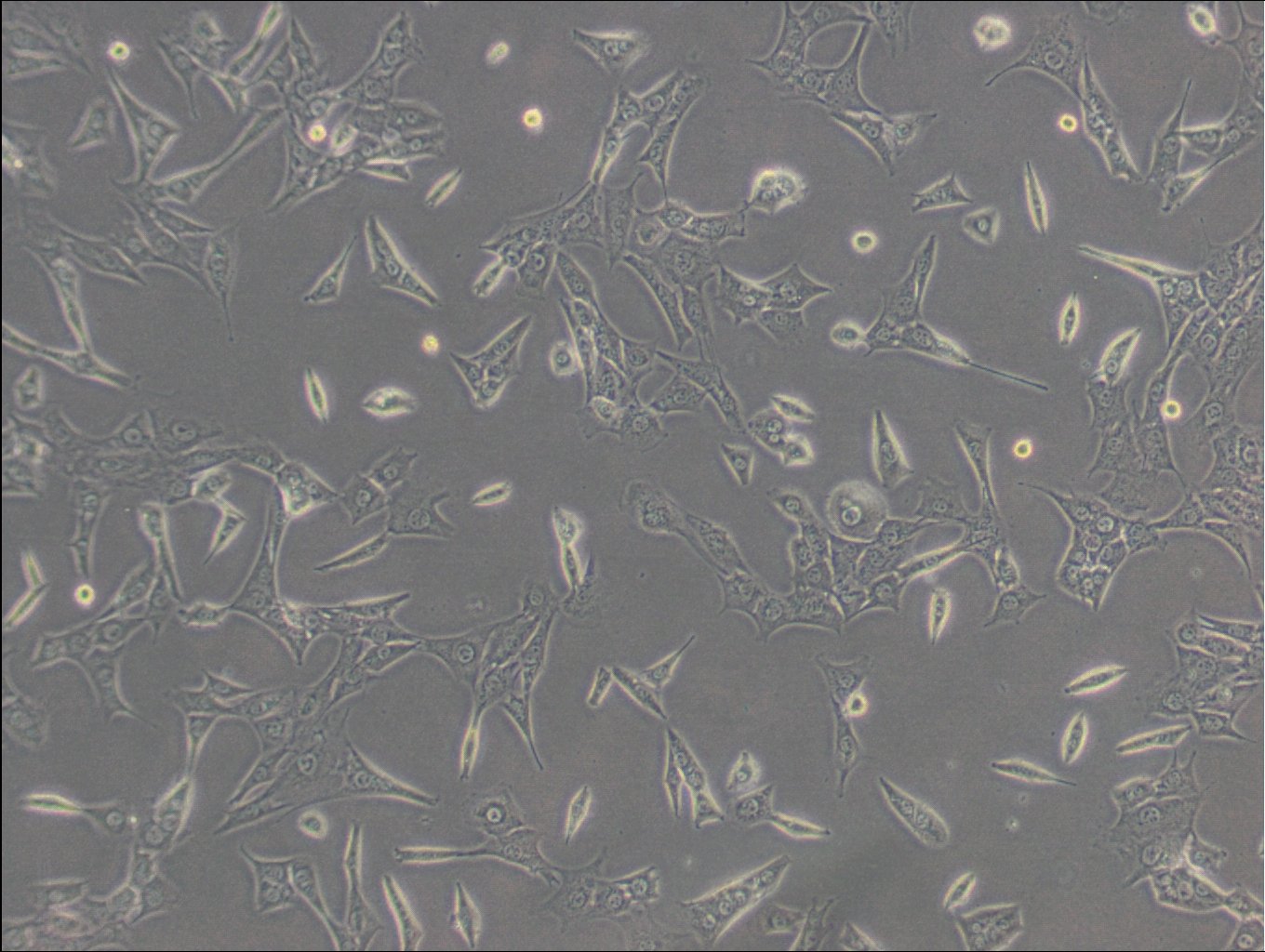
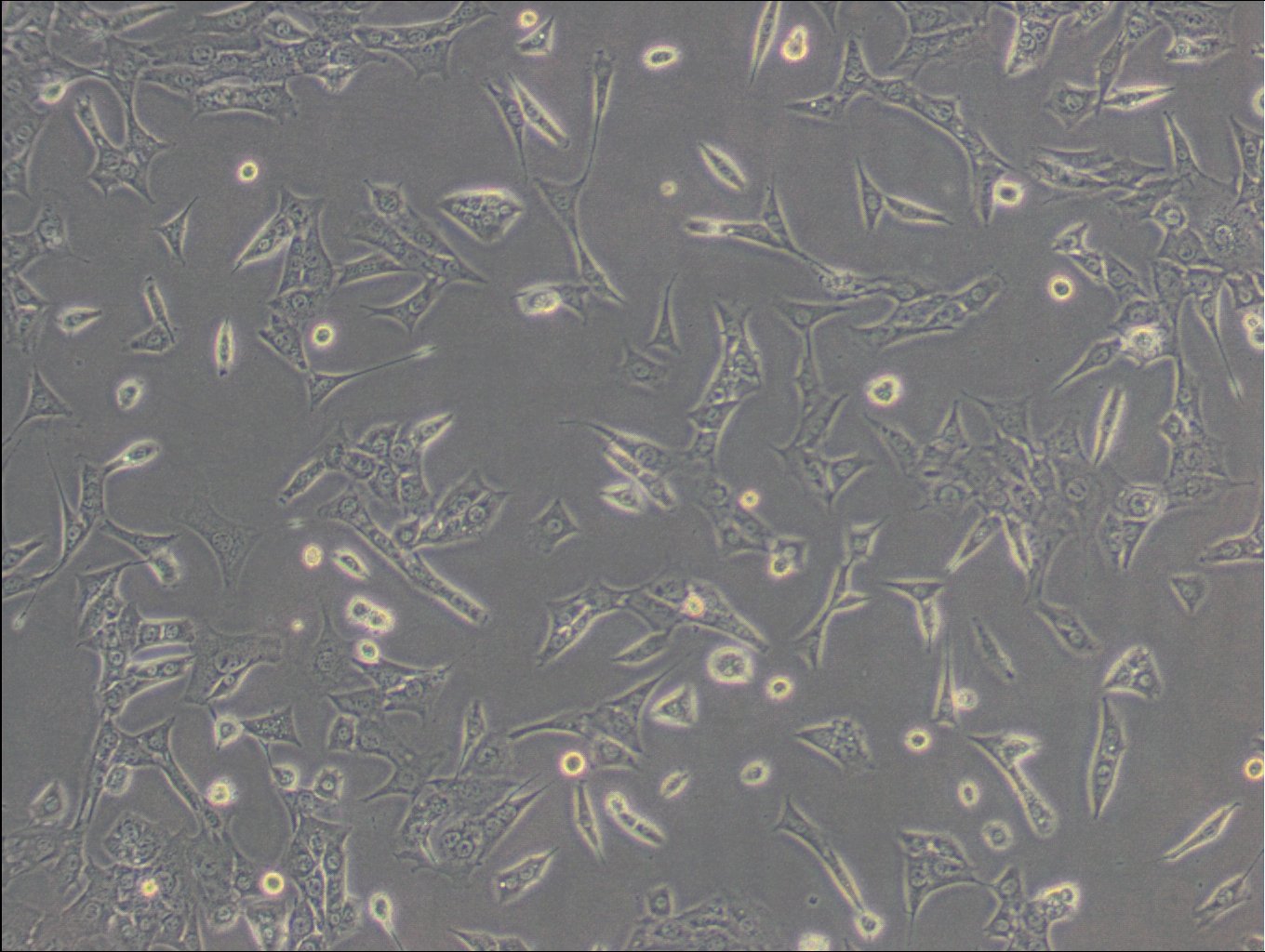
| Description | C127 cells, an unparalleled mammalian cell line, originate from murine mammary epithelial tissues. These cells provide a robust foundation for a diverse array of biological inquiries. These cells have been subjected to a meticulously designed procedure, involving infection with specially engineered viruses, leading to the integration of T7 RNA polymerase driven by a viral promoter. To further increase their adaptability, an additional recombinant virus carrying CFTR cDNA under the regulation of a T7 promoter, or a transfected plasmid with the T7 promoter, is introduced into the cells. C127 cells are epithelial, reflecting their extraction from mammary gland tissues, and grow adherent. C127 cells multiply rapidly and can be utilized to study cellular activities, expansion, and differentiation under a variety of testing conditions. The cells' unique genetic alterations make them a perfect candidate for protein expression studies by tailoring them to generate specific proteins. C127 cells also commonly used in the area of stable cell transfection. Scientists can implant foreign genetic material into these cells, facilitating the examination of gene functions, protein interactions, and the impacts of genetic alterations. Moreover, C127 cells have gained notoriety in the field of 3D cell culture. Using C127 cells in 3D culture offers researchers an opportunity to delve into the intricacies of cell-cell interactions, tissue formation, and disease modeling with increased physiological relevance. |
|---|---|
| Organism | Mouse |
| Tissue | Mammary gland |
| Disease | Malignant neoplasms of the mouse mammary gland |
| Synonyms | C-127 |
Characteristics
| Gender | Female |
|---|---|
| Morphology | Epithelial |
| Growth properties | Adherent |
Identifiers / Biosafety / Citation
| Citation | C127 (Cytion catalog number 305169) |
|---|---|
| Biosafety level | 1 |
Expression / Mutation
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:2 to 1:4 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | C127 cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
