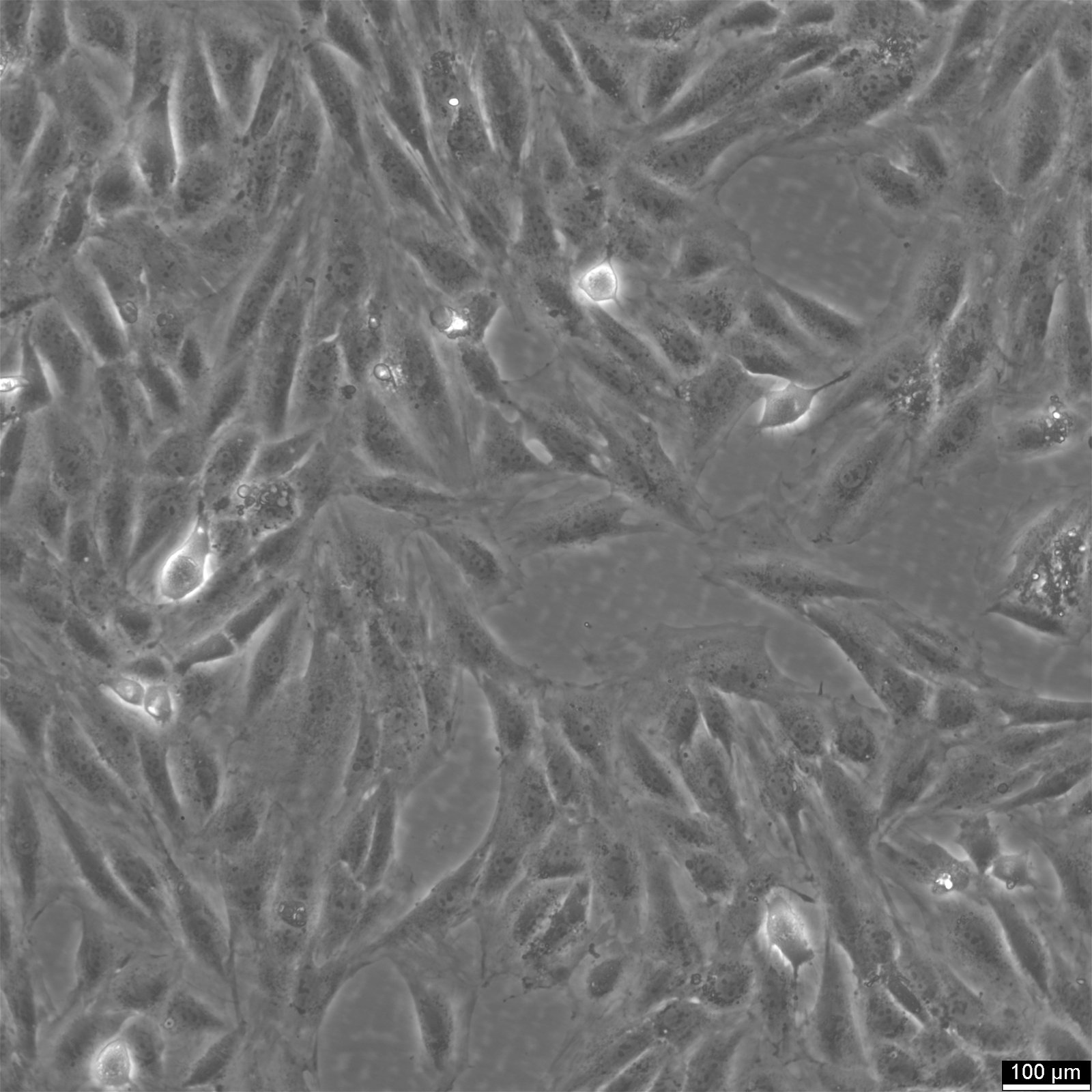
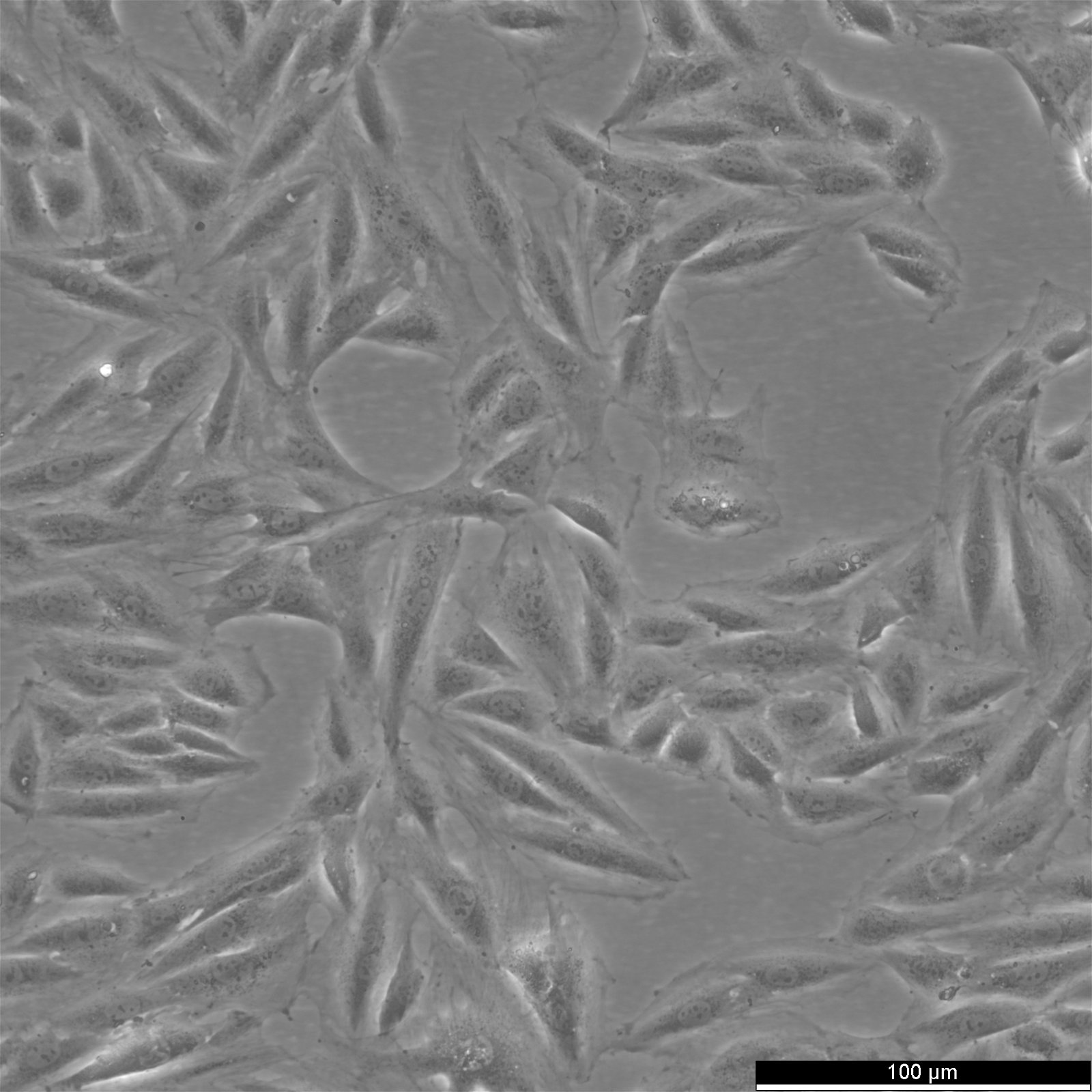
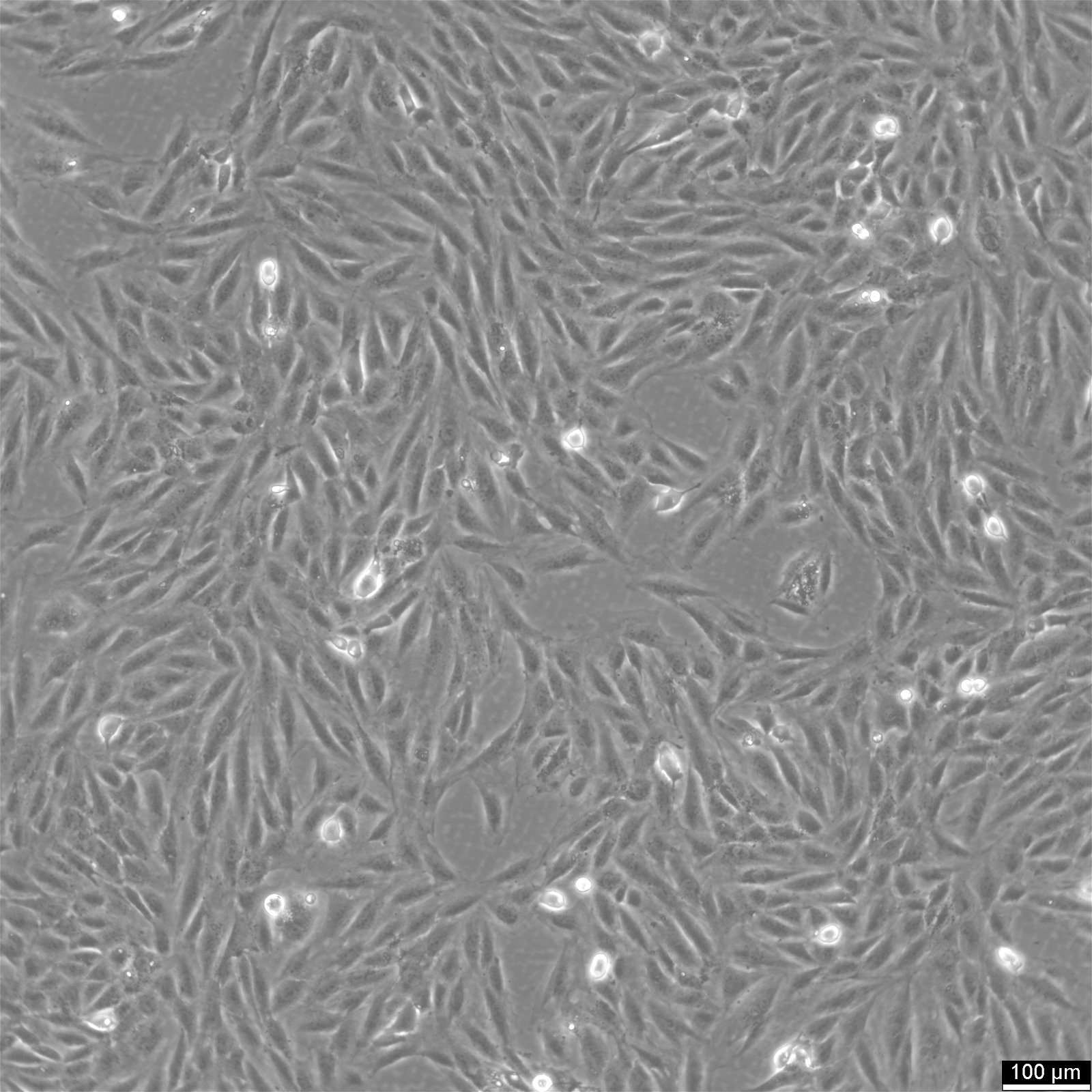
H9c2(2-1) Cells









Product number:
305203
General information
| Description | Introducing H9c2(2-1) Cells: A Versatile Cell Model for Cardiomyocyte Research H9c2(2-1) cells are a specialized subclone derived from the original clonal cell line obtained from embryonic BD1x rat heart tissue. These cells exhibit remarkable properties similar to skeletal muscle cells, making them an ideal model for studying cardiac muscle function. When stimulated by acetylcholine, the myoblastic cells in the H9c2(2-1) cell line can fuse and form multinucleated myotubes. These cells are derived from the embryonic heart tissue of the Rattus norvegicus (Rat) species, specifically the cardiac muscle. As an alternative to primary cardiomyocytes, H9c2 cells offer a valuable model for cardiac research. By reducing the serum concentration in the cell culture media and introducing all-trans-retinoic acid (RA), the H9c2 cells can be induced to differentiate into a cardiac-like phenotype. This differentiation process results in multinucleated cells with reduced proliferative capacity, resembling cardiomyocytes. In a groundbreaking study, the transcriptional analysis of H9c2 cells was conducted in two differentiation states: embryonic cells and differentiated cardiac-like cells. The findings revealed significant upregulation of genes encoding cardiac sarcomeric proteins, calcium transporters, and associated machinery. Notably, genes associated with mitochondrial energy production, such as respiratory chain complexes subunits, mitochondrial creatine kinase, carnitine palmitoyltransferase I, and uncoupling proteins, were also observed to increase expression during differentiation. Undifferentiated H9c2 myoblasts demonstrated increased expression of genes related to pro-survival proteins and cell cycle regulation. The differentiation of H9c2 cells led to the remodelling of mitochondrial function and increased transcripts and protein levels associated with calcium handling, glycolytic and mitochondrial metabolism. The involvement of PI3K, PDK1, and p-CREB in H9c2 differentiation was also identified. Complex analysis of differently expressed transcripts further revealed significant upregulation of genes associated with cardiac muscle contraction, dilated cardiomyopathy, and other pathways specific to cardiac tissue. The H9c2 (2–1) cell line offers several advantages over primary cardiomyocytes. Primary cardiomyocytes are delicate and challenging to maintain in culture for extended periods. Moreover, their isolation requires the sacrifice of laboratory animals, raising ethical concerns. In contrast, H9c2 cells provide a reliable in vitro model for studying cardiac and skeletal muscle, enabling cell biology, electrophysiology, and toxicology research without the need for animal sacrifice. Initially isolated from the ventricular part of a BDIx rat heart, the H9c2 cell line undergoes selective serial passages, allowing the separation of different components based on their adhesion kinetics. Although not fully differentiated into adult cardiomyocytes, these cells exhibit cardiomyocyte-specific markers, demonstrating their potential for cardiac research. By culturing H9c2 cells in a low serum concentration media, they can differentiate from mono-nucleated myoblasts to myotubes, assuming an elongated shape and parallel positioning. The addition of all-trans retinoic acid (RA) to the media further induces the presence of cells with an adult cardiac muscle phenotype characterized by the overexpression of the alpha-1 subunit of L-type calcium channels. It is important to note that H9c2 cells do not possess contractile activity, even in their differentiated state. Nevertheless, these cells respond similarly to various stimuli as isolated neonatal cardiomyocytes, including developing hypertrophic responses. However, it is crucial to consider the differentiation between state |
|---|---|
| Organism | Rat |
| Tissue | Heart, myocardium |
| Synonyms | H9c2 (2-1), H9c2, H9C2 |
Characteristics
| Age | Embryo |
|---|---|
| Morphology | Myoblast |
| Growth properties | Adherent |
Identifiers / Biosafety / Citation
| Citation | H9c2(2-1) (Cytion catalog number 305203) |
|---|---|
| Biosafety level | 1 |
Expression / Mutation
| Receptors expressed | Acetylcholine, expressed |
|---|---|
| Protein expression | Myokinase, Creatine Phosphokinase, Myosin |
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:2 to 1:4 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | H9c2(2-1) cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|
