Hey Cells













General information
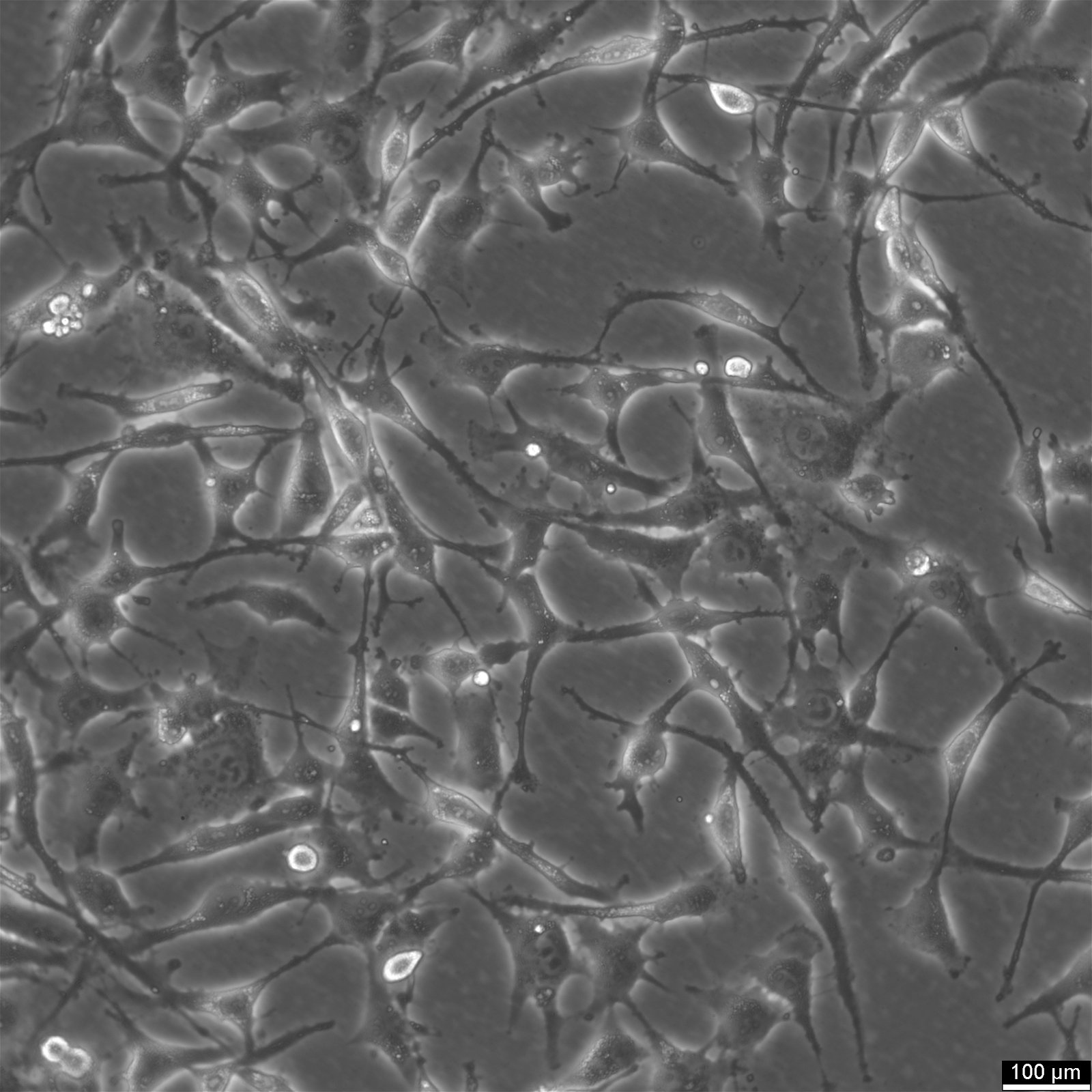
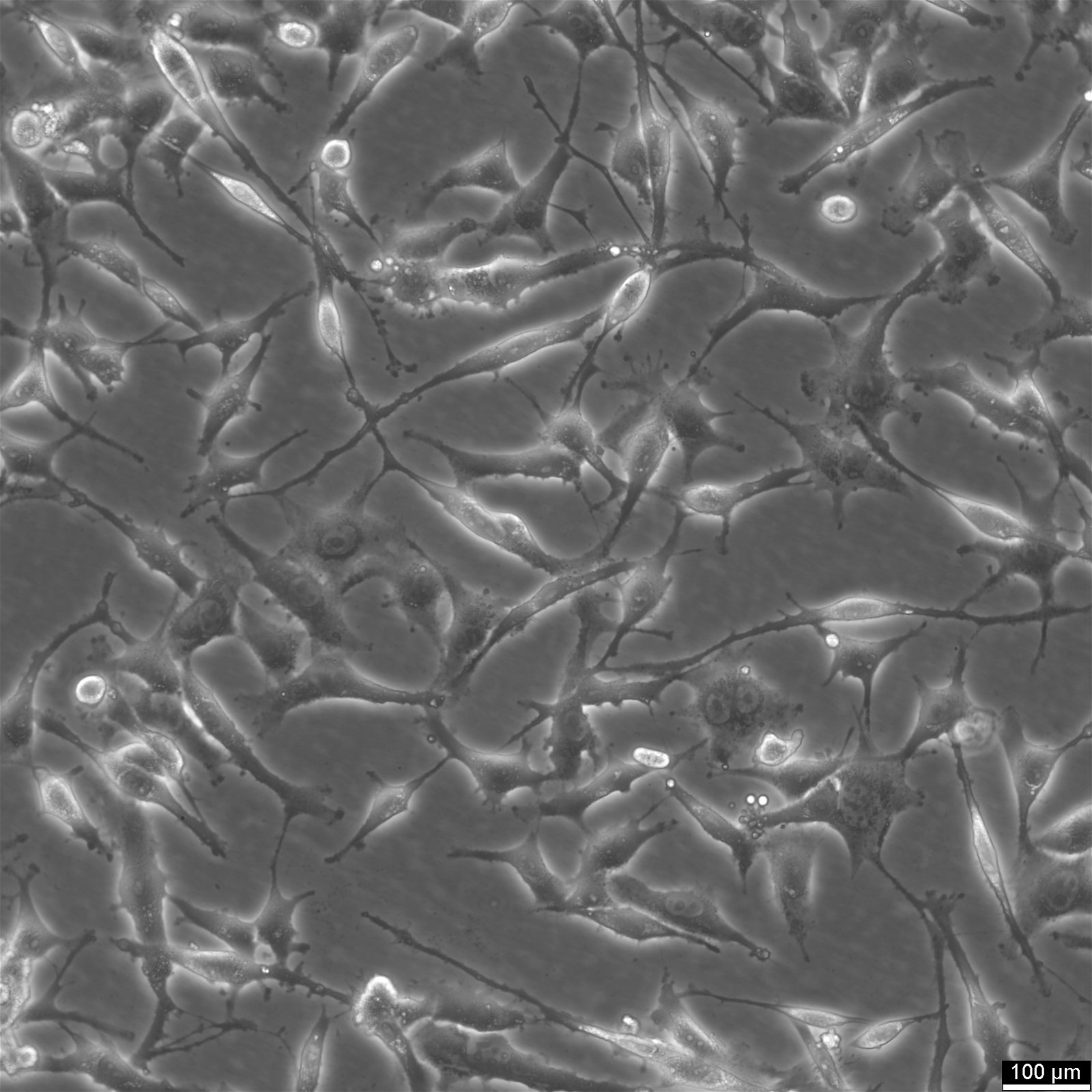
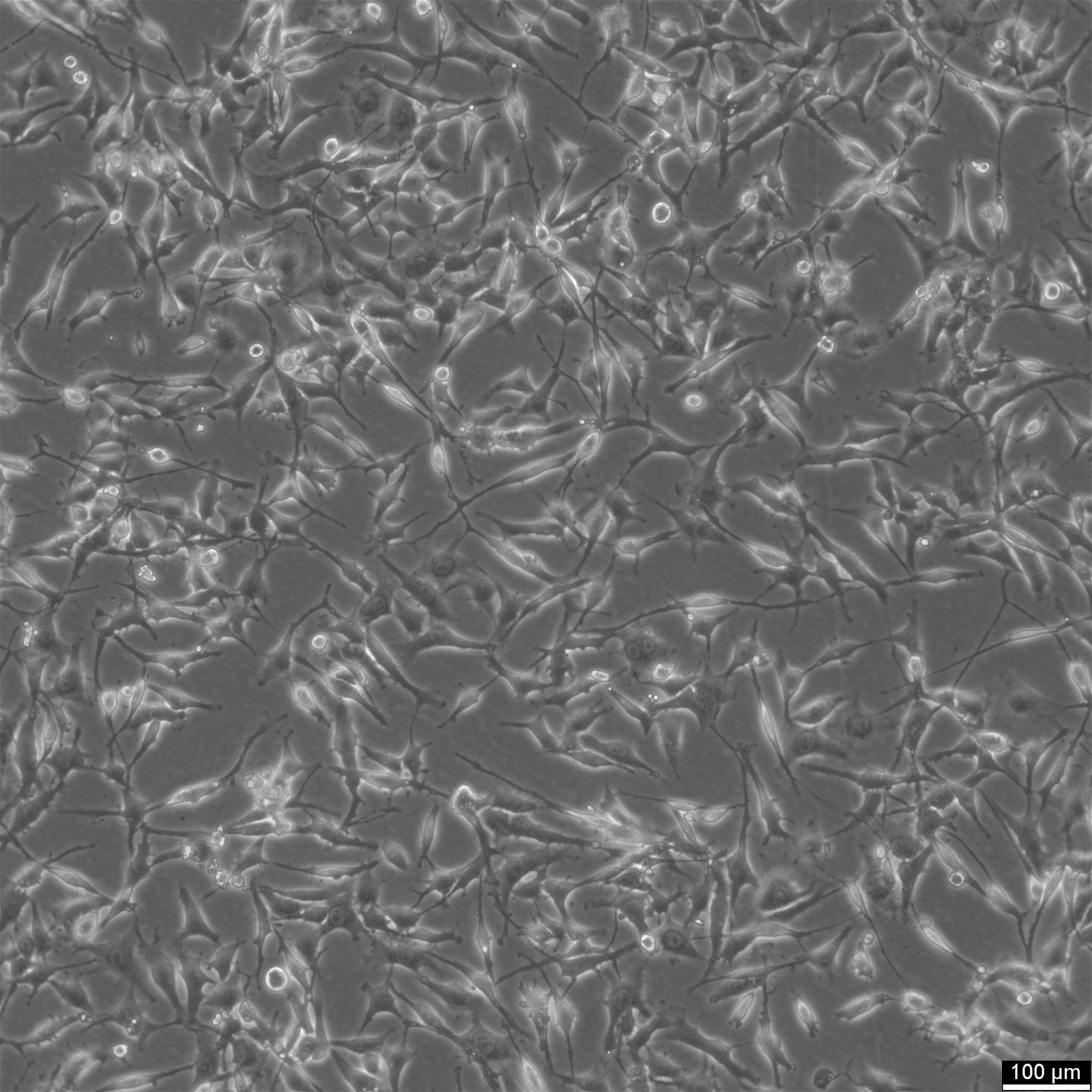
| Description | HEY Cells, derived from a human ovarian cancer xenograft, are a valuable resource for cancer researchers seeking to advance their understanding of papillary cystadenocarcinoma, a moderately differentiated form of ovarian cancer. The parental cell line, HEY, was initially obtained from a peritoneal sample of a Caucasian patient diagnosed with this specific type of cancer. A derivative known as HEY-T30 (CRL-3252) was developed through a carefully designed process to enhance the versatility and usefulness of HEY Cells. This involved subjecting the parental HEY cell line to stepwise escalating concentrations of Taxol for six months. HEY-T30 is now maintained in a medium containing 30 nmol/L Taxol, ensuring its stability and consistency. It is worth noting that Jurriaan Brouwer-Visser Institution is the designated repository for this cell line. HEY, Cells are particularly well-suited for applications involving 3D cell culture, a technique that more closely mimics the physiological environment of tumours. Their ability to grow in semisolid culture and as xenografts in immunologically deprived CBA/CJ mice highlights their adaptability and potential for in vivo studies. By incorporating HEY Cells into cancer research, scientists can uncover crucial insights into the development and progression of papillary cystadenocarcinoma. These cells are invaluable for exploring novel therapeutic strategies, identifying potential drug targets, and evaluating treatment efficacy. In summary, HEY Cells provide researchers with a robust and reliable resource for investigating ovarian cancer. With their origins in a patient sample and their epithelial-like morphology, these cells faithfully replicate key characteristics of papillary cystadenocarcinoma. Their applications in 3D cell culture and cancer research make them essential in advancing our understanding of this challenging disease. |
|---|---|
| Organism | Human |
| Tissue | Ovary |
| Disease | High grade ovarian serous adenocarcinoma |
| Synonyms | HEY |
Characteristics
| Age | Unspecified |
|---|---|
| Gender | Female |
| Ethnicity | European |
| Morphology | Epithelial |
| Growth properties | Adherent |
Identifiers / Biosafety / Citation
| Citation | Hey (Cytion catalog number 305017) |
|---|
Expression / Mutation
| Tumorigenic | Yes |
|---|
Handling
| Culture Medium | DMEM, w: 4.5 g/L Glucose, w: 4 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: 1.0 mM Sodium pyruvate (Cytion article number 820300a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Doubling time | 20 to 30 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:3 to 1:5 |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures |
|
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|---|
| STR profile |
Amelogenin: x,x
CSF1PO: 10,11
D13S317: 11
D16S539: 8,12
D5S818: 11,12
D7S820: 12
TH01: 8,9.3
TPOX: 11
vWA: 16,17
D3S1358: 16
D21S11: 30
D18S51: 15
Penta E: 7,13
Penta D: 9,13
D8S1179: 13
FGA: 20,21
D6S1043: 11,12
D2S1338: 24,25
D12S391: 17,22
D19S433: 13,14
|
