KYSE-30 Cells
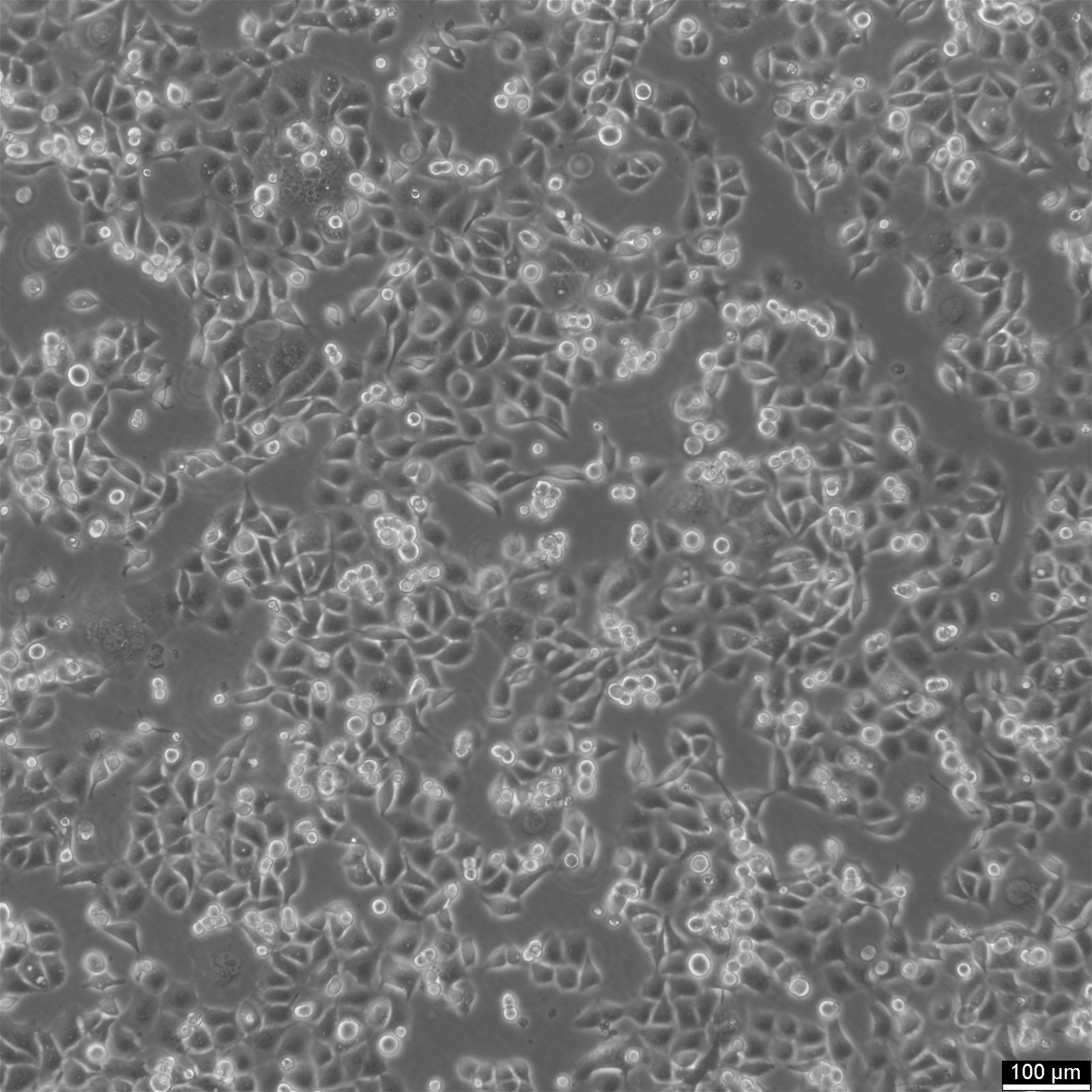

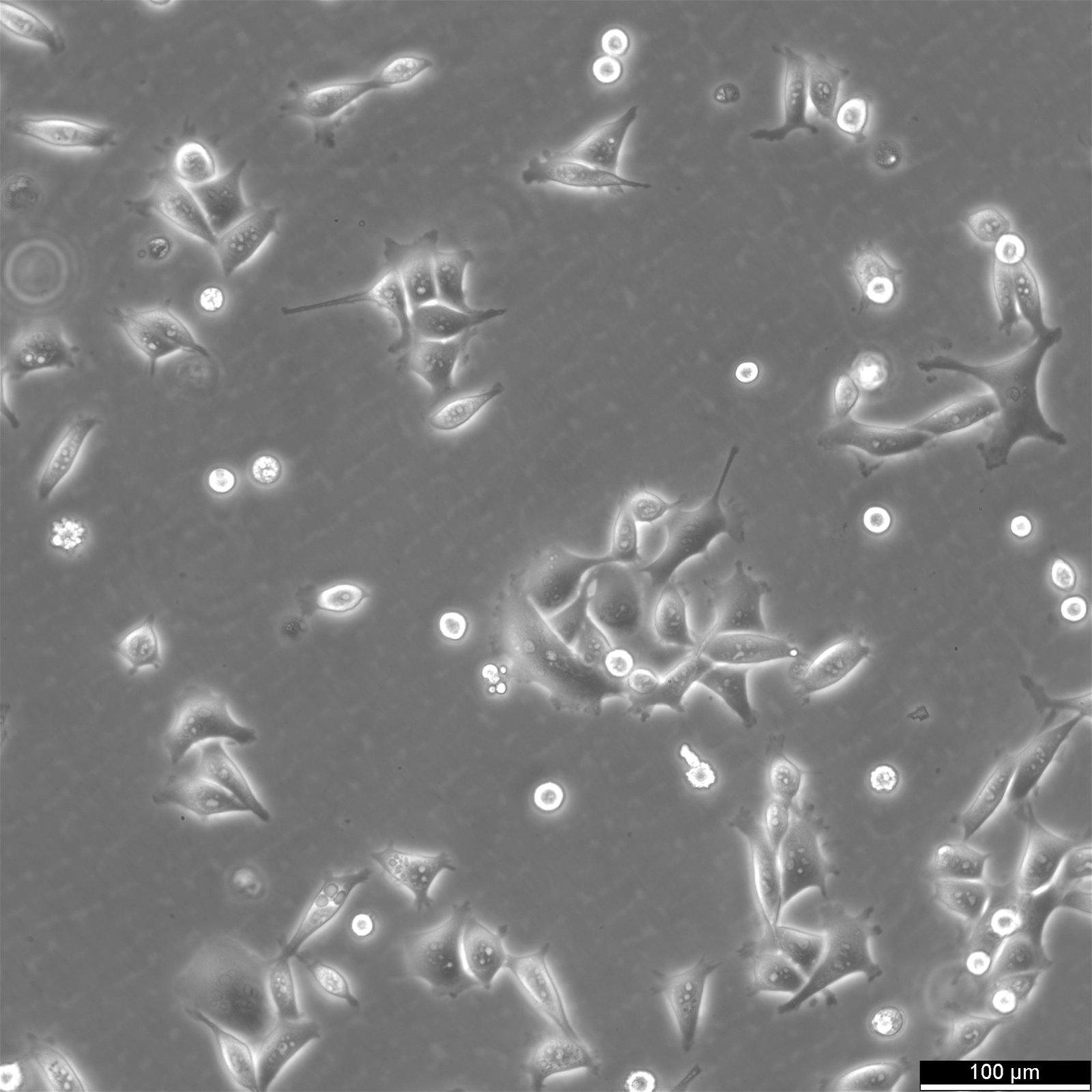
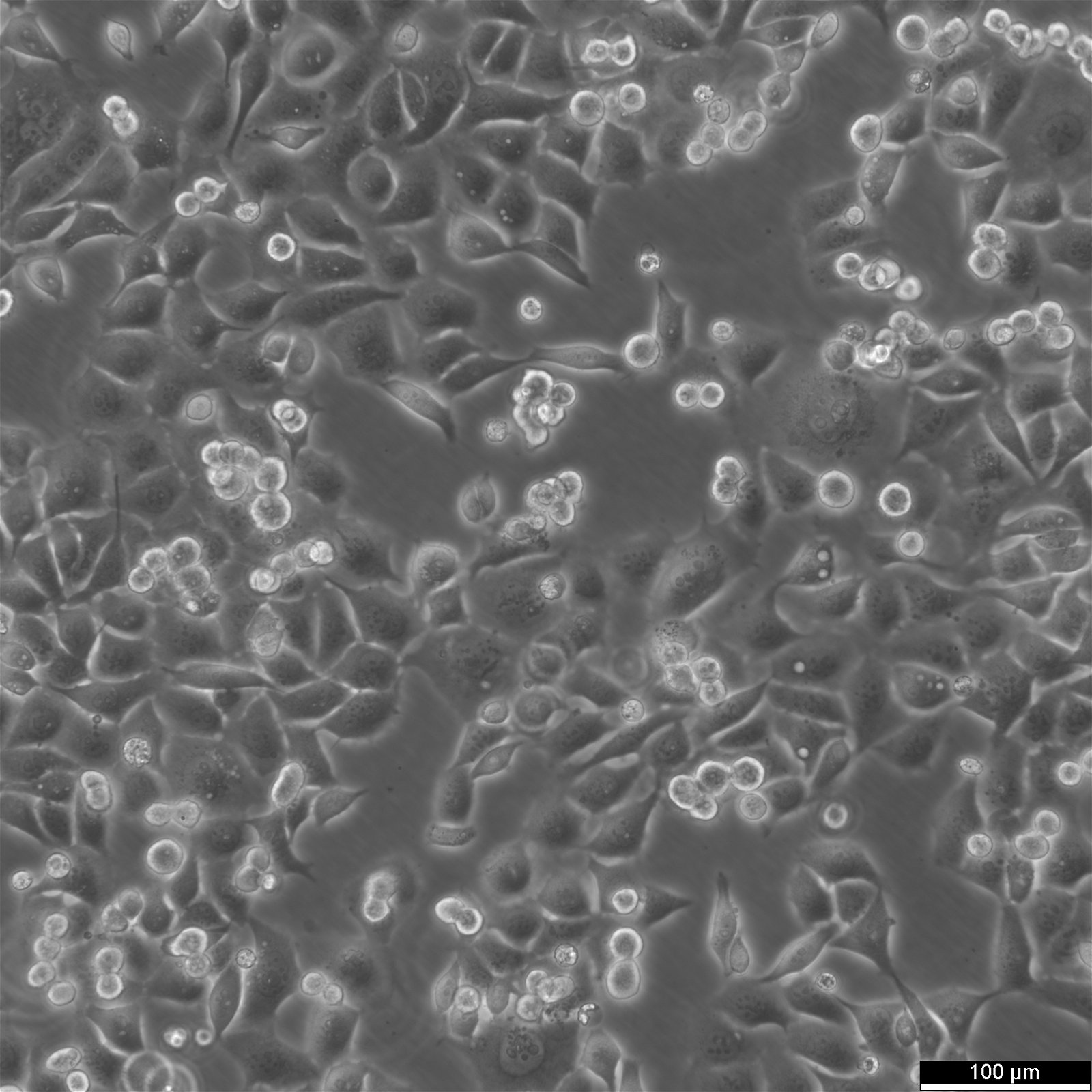
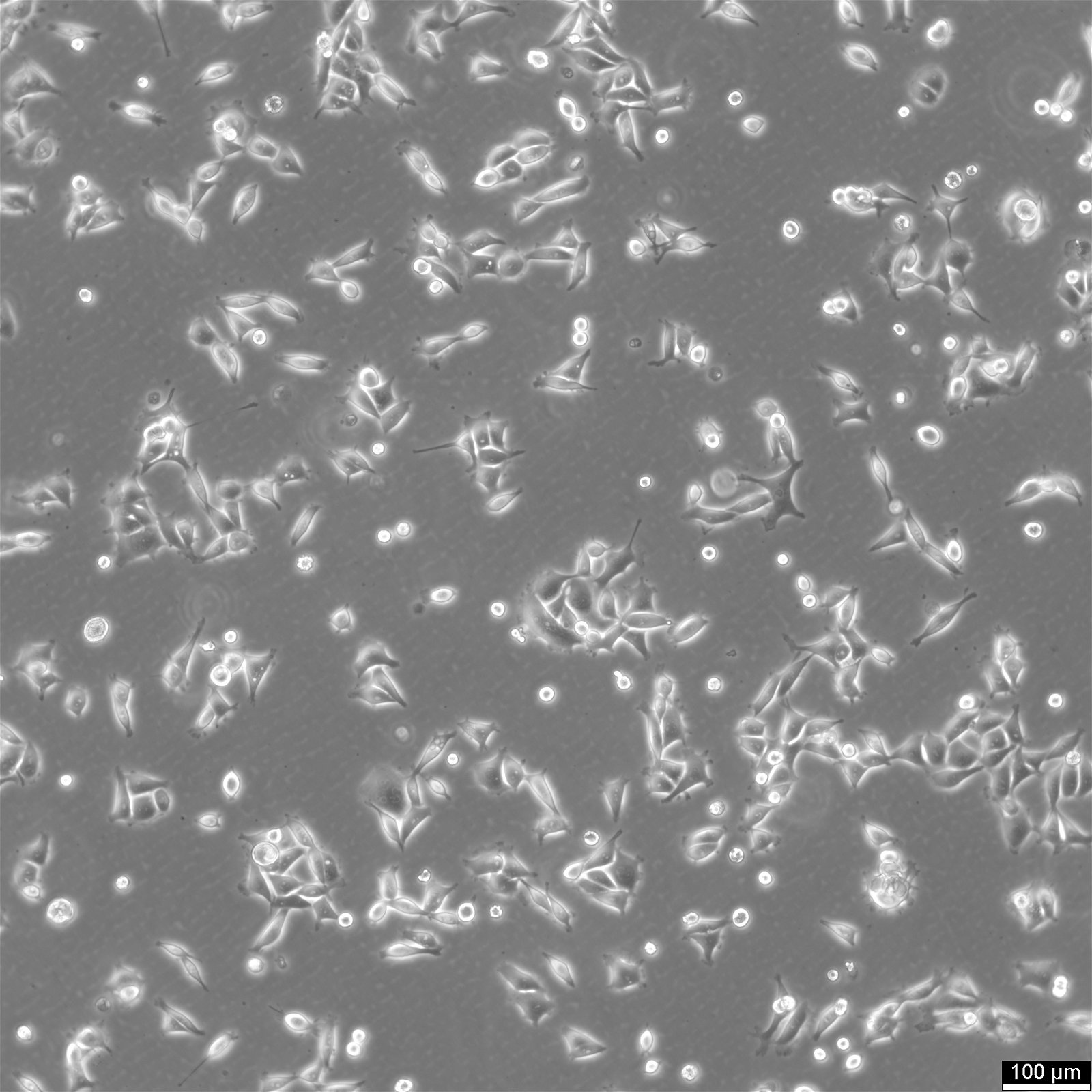















General information
| Description | This cell line was established from the mucosal surface of a well differentiated invasive esophageal squamous cell carcinoma from a 64-year-old Japanese male prior to treatment. The cell line was used for heterotransplanting tumours to athymic mice. The cells have a p53 mutation at the splice acceptor site and amplification of cERB B, MYC and CYCLIN D1. |
|---|---|
| Organism | Human |
| Tissue | Esophageal Squamous Epithelium |
| Disease | Esophageal squamous cell carcinoma |
| Synonyms | Kyse-30, KYSE 30, KYSE30, Kyse30, KYSE0030 |
Characteristics
| Age | 64 years |
|---|---|
| Gender | Male |
| Ethnicity | Asian |
| Morphology | Epithelial-Like, With Long Pseudopod |
| Growth properties | Adherent |
Identifiers / Biosafety / Citation
| Citation | KYSE-30 (Cytion catalog number 305094) |
|---|---|
| Biosafety level | 1 |
Expression / Mutation
Handling
| Culture Medium | Please mix Ham's F12 and RPMI 1640 in a 50:50 ratio (Cytion article numbers 820600a and 820702a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Doubling time | 20 to 30 hours |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1: 3 to 1: 5 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures |
|
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|---|
| STR profile |
Amelogenin: x,x
CSF1PO: 10
D13S317: 9
D16S539: 10,12
D5S818: 11
D7S820: 11,11.3
TH01: 9
TPOX: 9
vWA: 16,18,19
D3S1358: 15,16
D21S11: 28
D18S51: 14
Penta E: 13
Penta D: 12
D8S1179: 12,15
FGA: 24
D6S1043: 11,20
D2S1338: 23
D12S391: 17,19
D19S433: 14.2,15.2
|
