SK-N-BE(2) Cells









Product number:
305058
General information
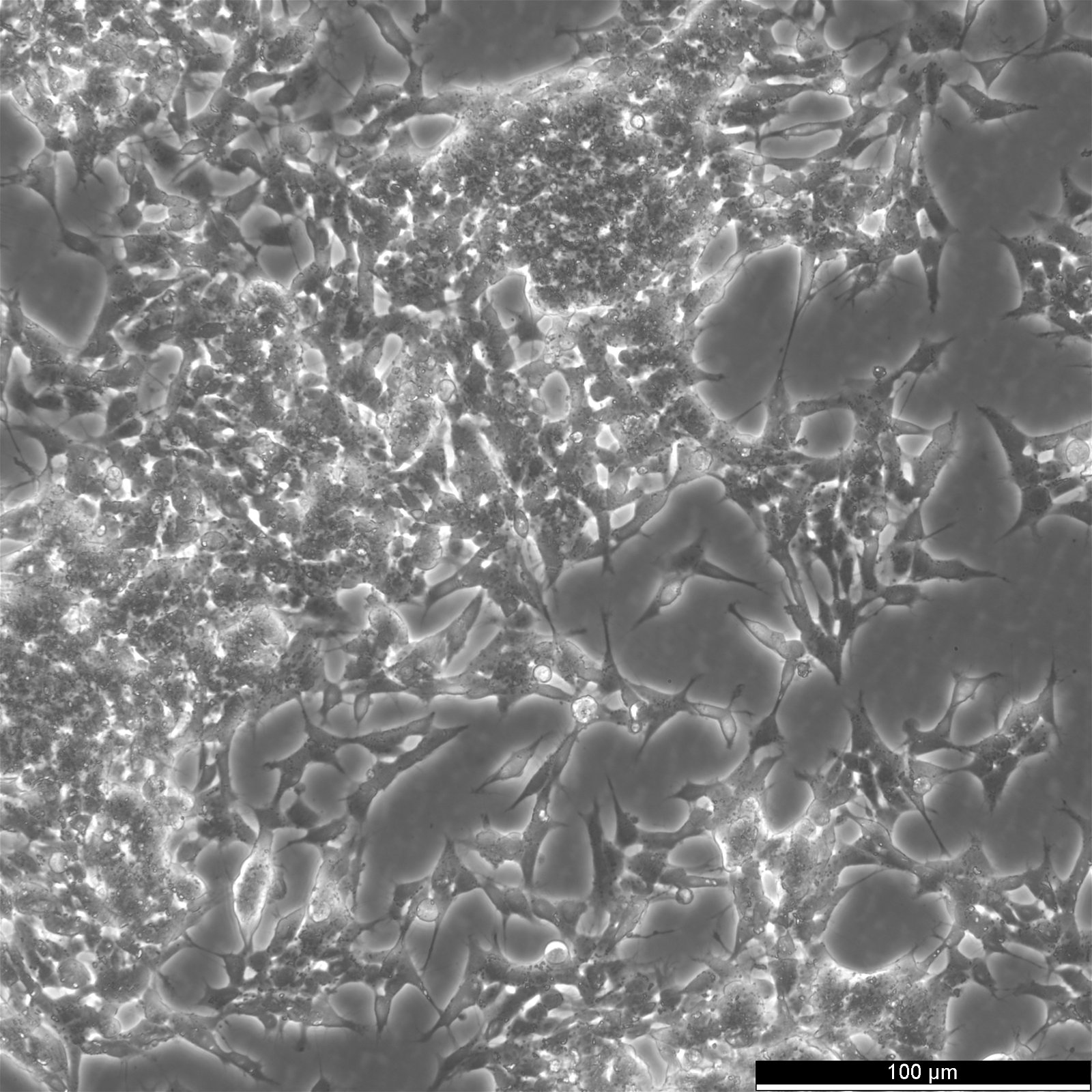
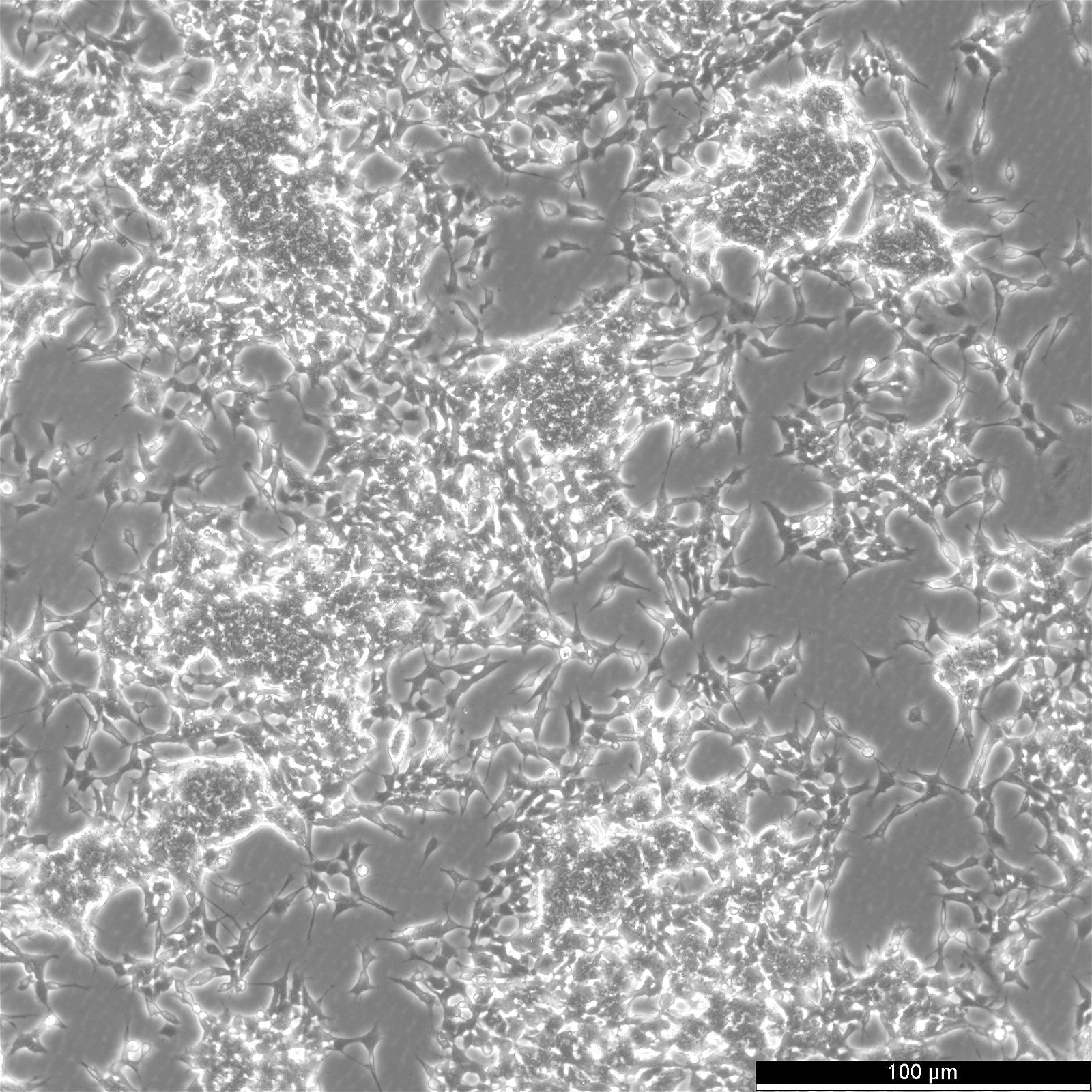
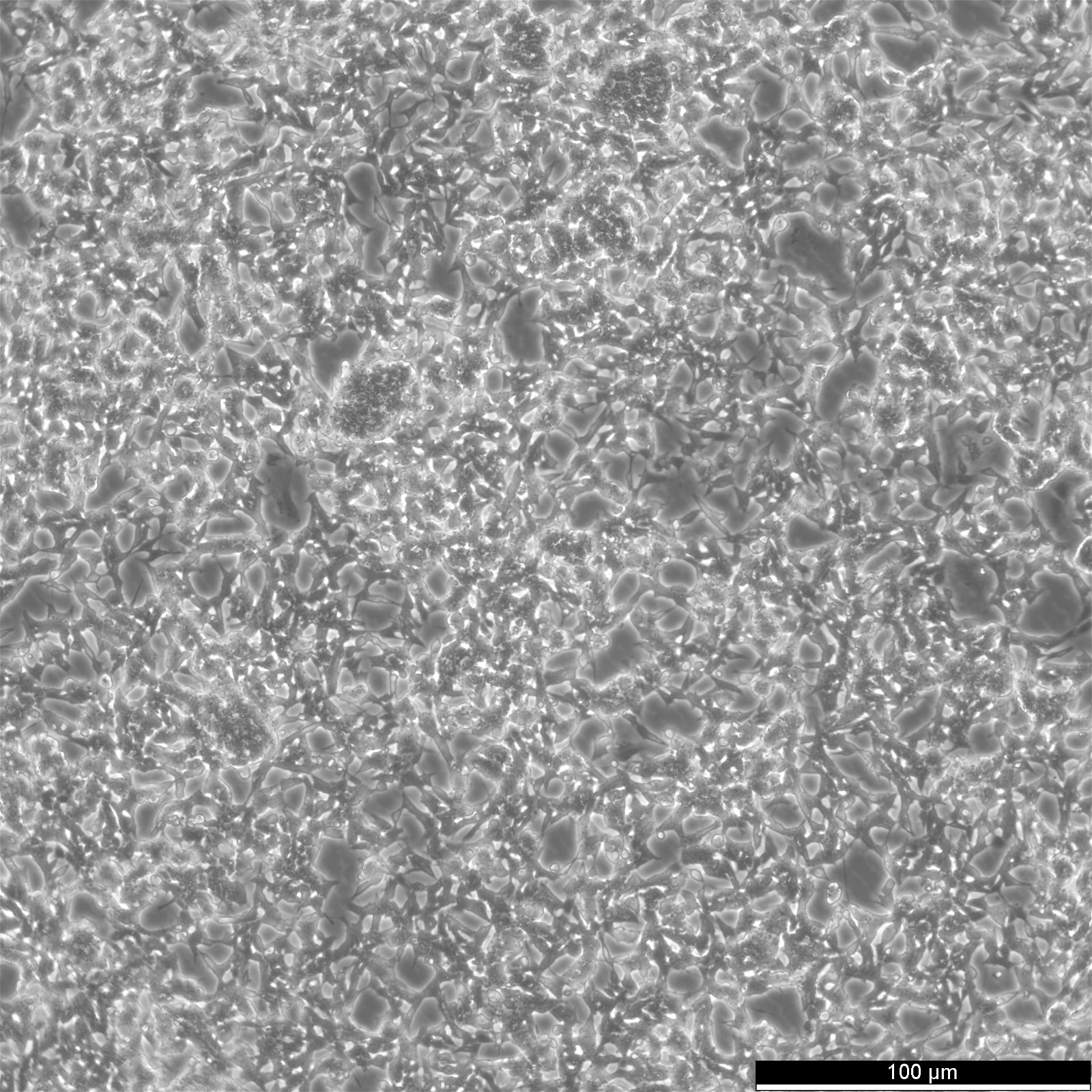
| Description | The cells exhibit moderate levels of dopamine beta hydroxylase activity.SK-N-BE(2) cells have a reported saturation density greater than 1×10^6 cells/cm^2.The morphology of the cells varies with some cells having long processes and others that are epithelioid like.The cells will aggregate, form clumps and float. |
|---|---|
| Organism | Human |
| Tissue | Brain |
| Disease | Neuroblastoma |
| Metastatic site | Bone marrow |
| Synonyms | SK-N-BE2, SK-N-BE-2, SKNBE(2), SKNBE-2, SKNBE2, SK-N-BE, SKNBE |
Characteristics
| Age | 2 years |
|---|---|
| Gender | Male |
| Ethnicity | European |
| Morphology | Neuroblast |
| Growth properties | Adherent/suspension |
Identifiers / Biosafety / Citation
| Citation | SK-N-BE(2) (Cytion catalog number 305058) |
|---|---|
| Biosafety level | 1 |
Expression / Mutation
| Tumorigenic | Yes |
|---|
Handling
| Culture Medium | Please mix EMEM and Ham's F12 in a 50:50 ratio (Cytion article numbers 820100c and 820600a) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Gather the suspension cells in a 15 ml tube and gently wash the adherent cells with PBS lacking calcium and magnesium (use 3-5 ml for T25 flasks and 5-10 ml for T75 flasks). Apply Accutase (1-2 ml for T25 flasks, 2.5 ml for T75 flasks) ensuring full coverage of the cell layer. Allow the cells to incubate at room temperature for 10 minutes. Following incubation, combine and centrifuge both the suspension and adherent cells. After centrifugation, carefully resuspend the cell pellet and transfer the cell suspension into new flasks containing fresh medium. |
| Split ratio | 1:2 to 1:4 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | SK-N-BE(2) cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|---|
| STR profile |
Amelogenin: x,y
CSF1PO: 10
D13S317: 11
D16S539: 9,11
D5S818: 12
D7S820: 9,10
TH01: 6,7
TPOX: 8,11
vWA: 18
D3S1358: 19
D21S11: 30,32.2
D18S51: 16
Penta E: 14,18
Penta D: 13,14
D8S1179: 13,14
FGA: 22,25
D6S1043: 11,19
D2S1338: 17,23
D12S391: 18,24
D19S433: 12,13
|
