SiHa Cells












General information
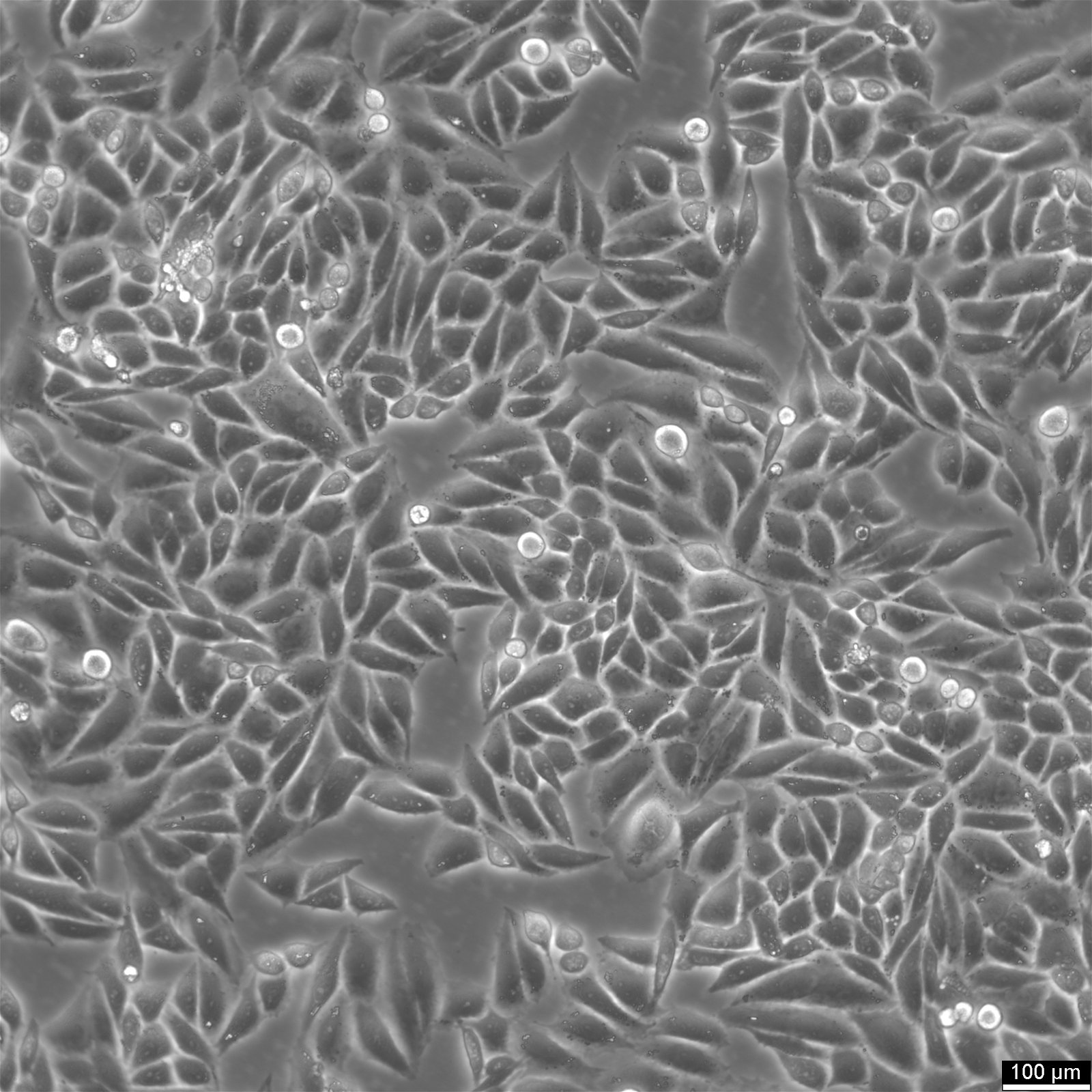
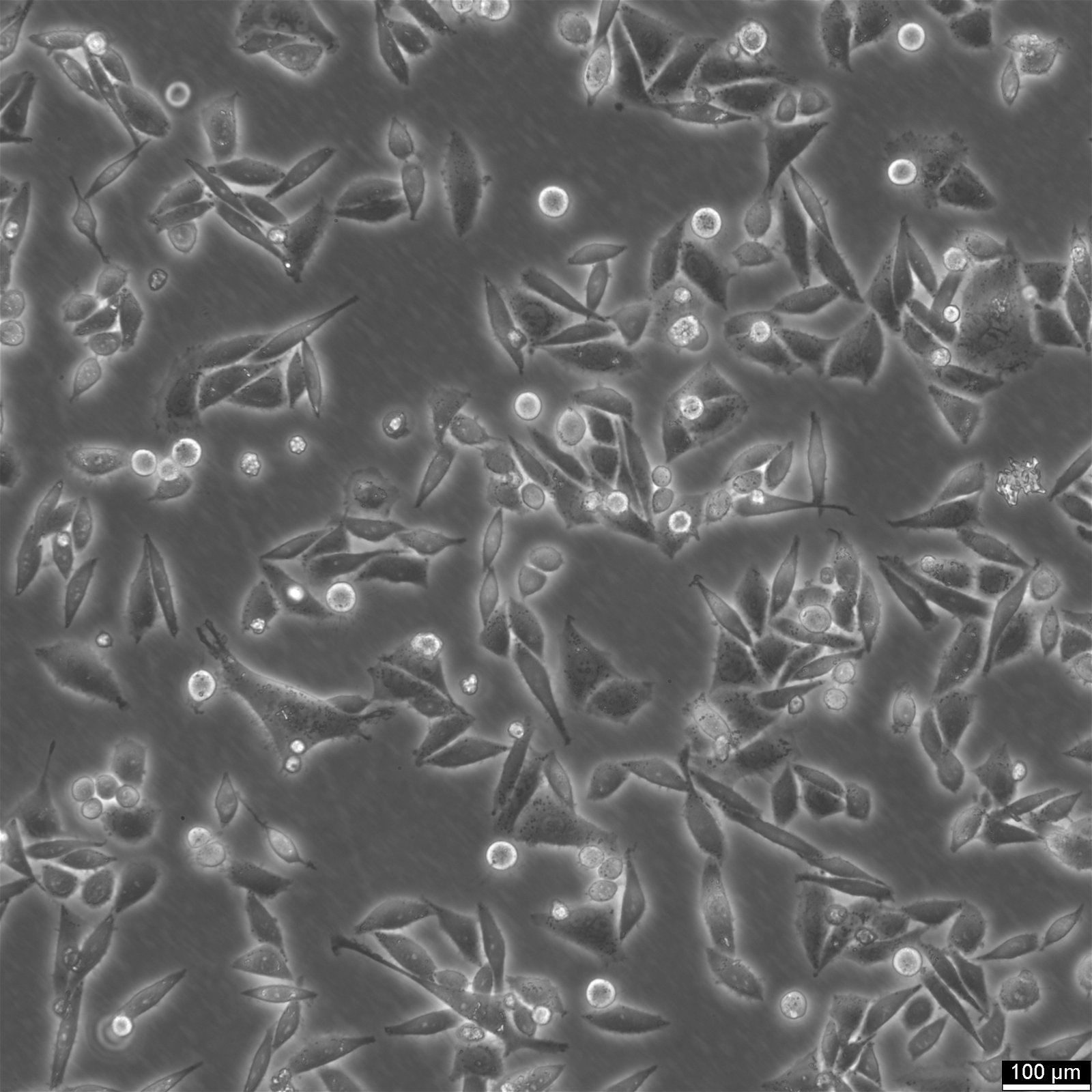
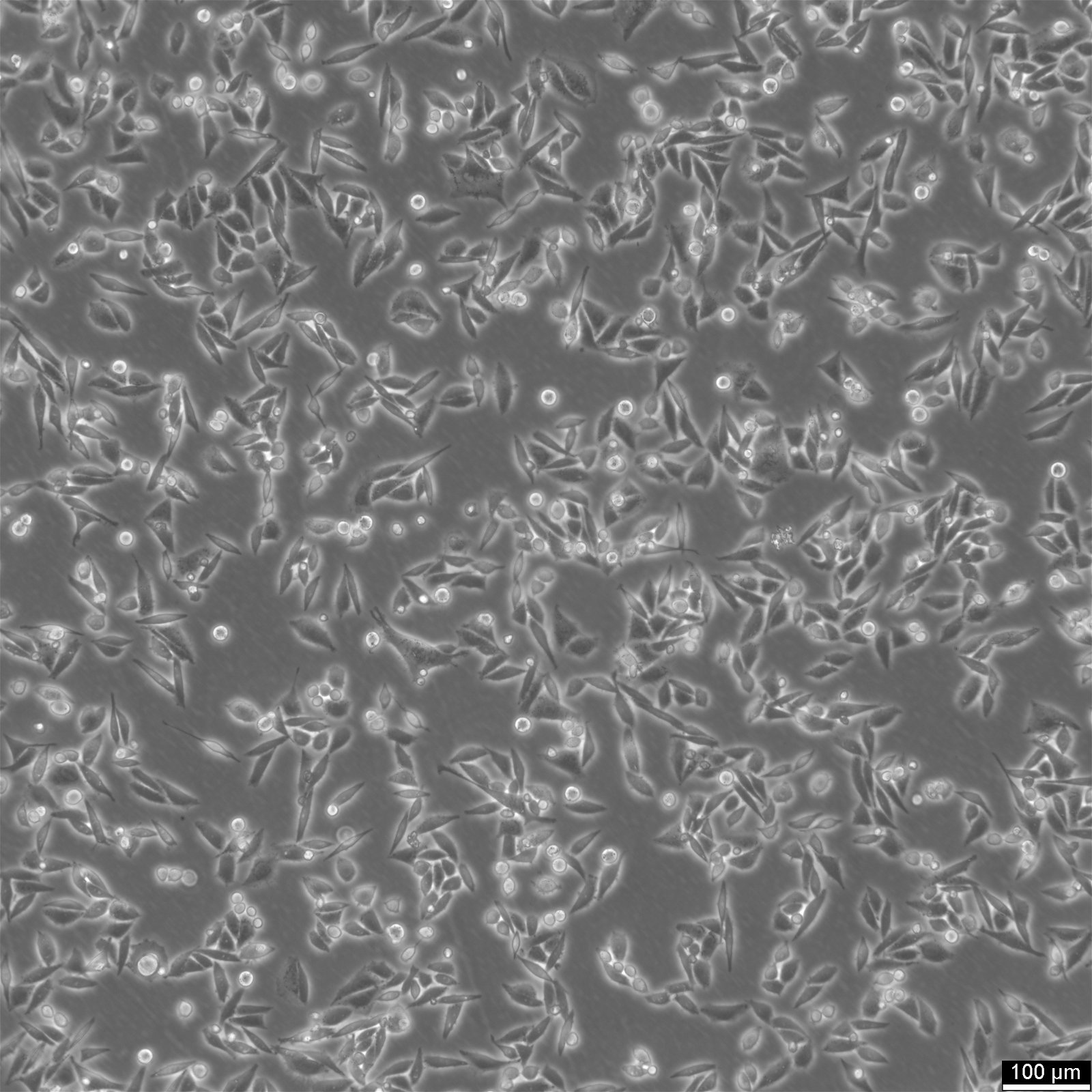
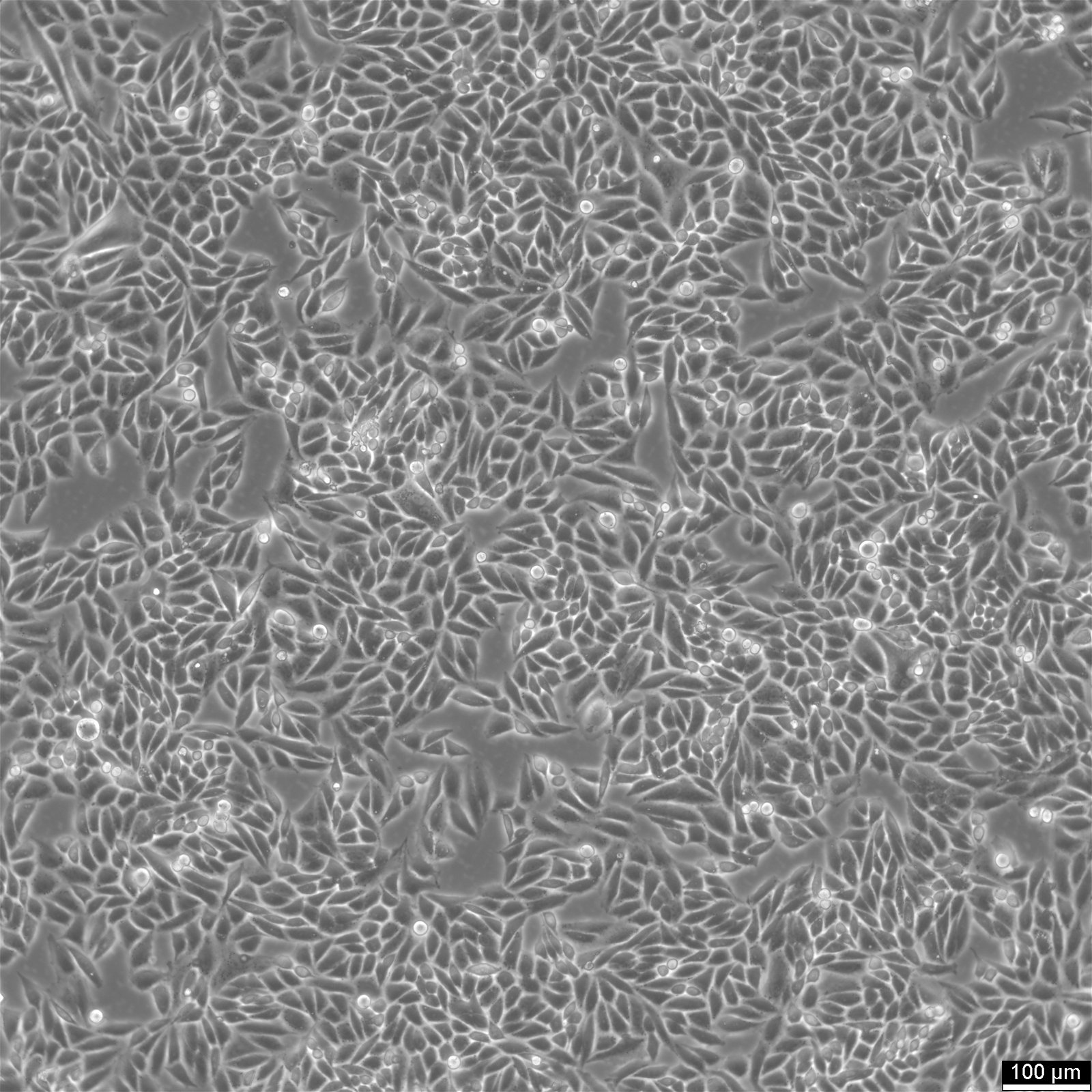
| Description | SiHa cells are a human cervical squamous cell carcinoma cell line that has been widely used in research for several decades. They were isolated from primary uterine biopsy fragments from a 55-year-old female Japanese patient with squamous cell carcinoma. This cell line is of great interest to researchers studying cervical cancer and other related diseases due to their unique genetic characteristics. SiHa cells have been found to express the p53+ and pRB+ genes, which are involved in cell cycle regulation, DNA repair, and tumor suppression. These genes make SiHa cells an ideal model for studying the molecular mechanisms of cancer development and progression. Additionally, SiHa cells are a suitable transfection host, making them an excellent tool for gene expression studies. SiHa cells have a hypertriploid karyotype, with an average chromosome number between 69 and 72. The SiHa cells are HPV-16 positive, showing integration of 1 to 2 copies of the viral genome per cell. Cells are tumorigenic, forming poorly differentiated epidermoid carcinoma (grade III) in nude mice. This makes them an excellent model for studying cancer progression and testing anti-cancer drugs. SiHa cell line expresses various isoenzymes, including AK-1, ES-D, G6PD, GLO-I, Me-2, PGM1, and PGM3. Electron microscopy revealed abundant tonofilaments in the cytoplasm and desmosomes at the cell junctions. The growth properties of SiHa cells are adherent, with a doubling time of 17 hours in 10% FBS media and 21 hours in 5% FBS media. Epithelial cell adhesion molecule (EpCAM) expression is present in 92% of SiHa cells, indicating their epithelial origin. They show strong cytokeratin expression but no vimentin expression. |
|---|---|
| Organism | Human |
| Tissue | Cervix |
| Disease | Human papillomavirus-related cervical squamous cell carcinoma |
| Synonyms | Siha, SIHA |
Characteristics
| Age | 55 years |
|---|---|
| Gender | Female |
| Ethnicity | Asian |
| Morphology | Epithelial |
| Growth properties | Adherent |
Identifiers / Biosafety / Citation
| Citation | SiHa (Cytion catalog number 305023) |
|---|---|
| Biosafety level | 1 |
Expression / Mutation
| Tumorigenic | Yes |
|---|
Handling
| Culture Medium | EMEM, w: 2 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: EBSS, w: 1 mM Sodium pyruvate, w: NEAA (Cytion article number 820100c) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Split ratio | 1:2 to 1:4 |
| Fluid renewal | 2 to 3 times per week |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | SiHa cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality control / Genetic profile / HLA
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|---|
| STR profile |
Amelogenin: x,x
CSF1PO: 12
D13S317: 11
D16S539: 12
D5S818: 9
D7S820: 10
TH01: 6,9
TPOX: 8
vWA: 14,17
D3S1358: 16,17
D21S11: 31
D18S51: 15
Penta E: 10,12
Penta D: 9
D8S1179: 13,16
FGA: 21
D6S1043: 18
D2S1338: 24
D12S391: 19,22
D19S433: 14.2
|
