WI-38 Cells
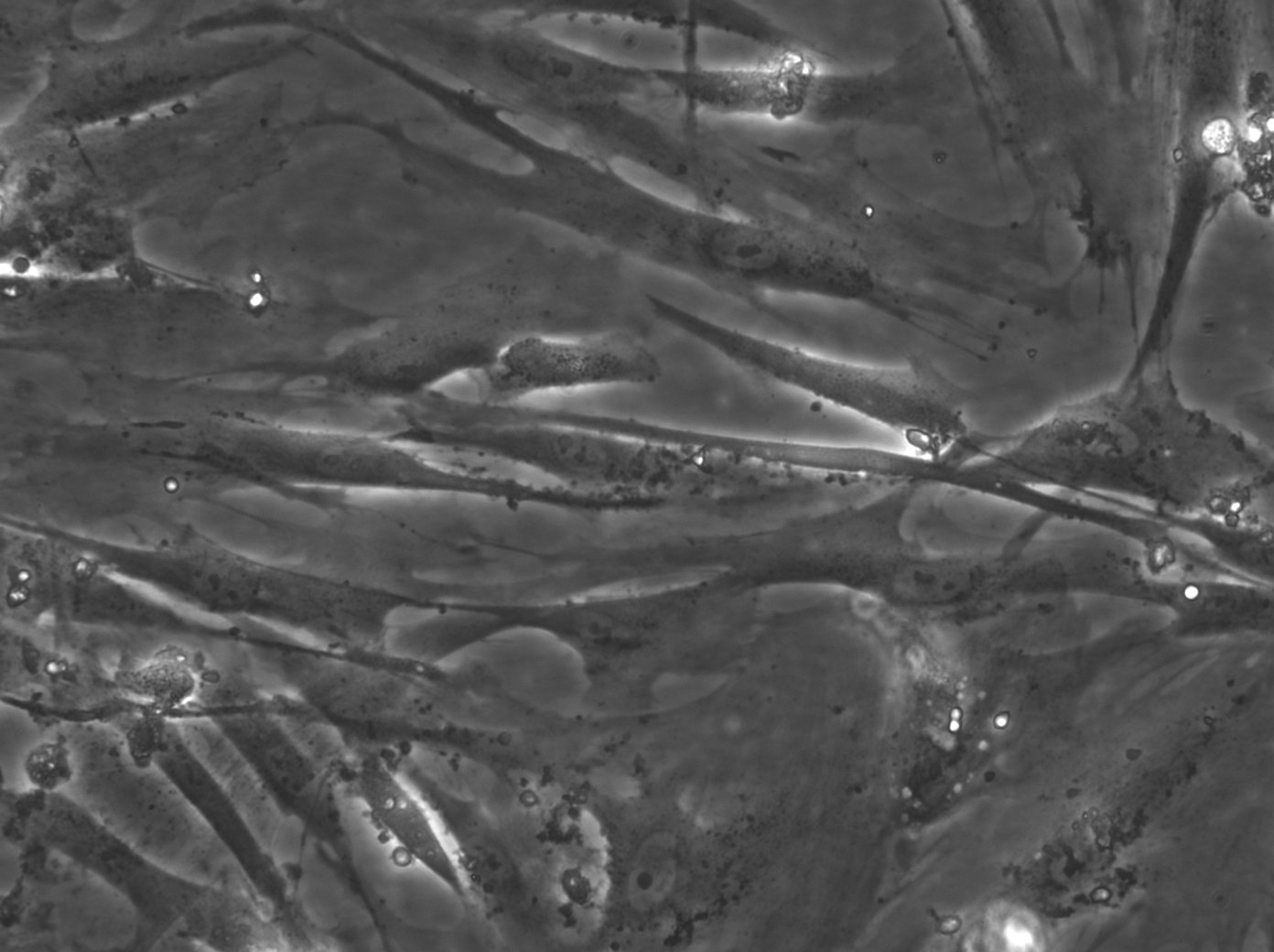
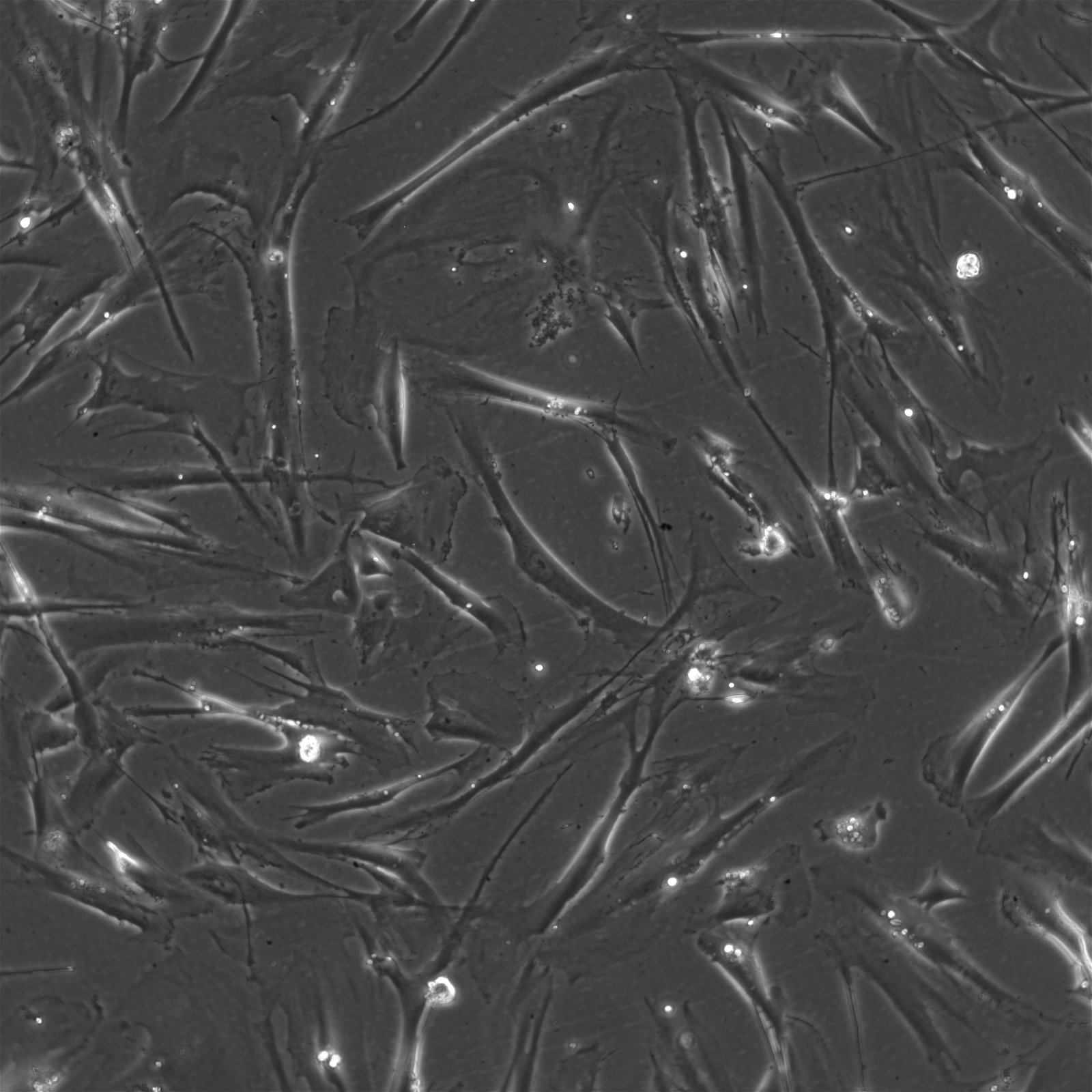
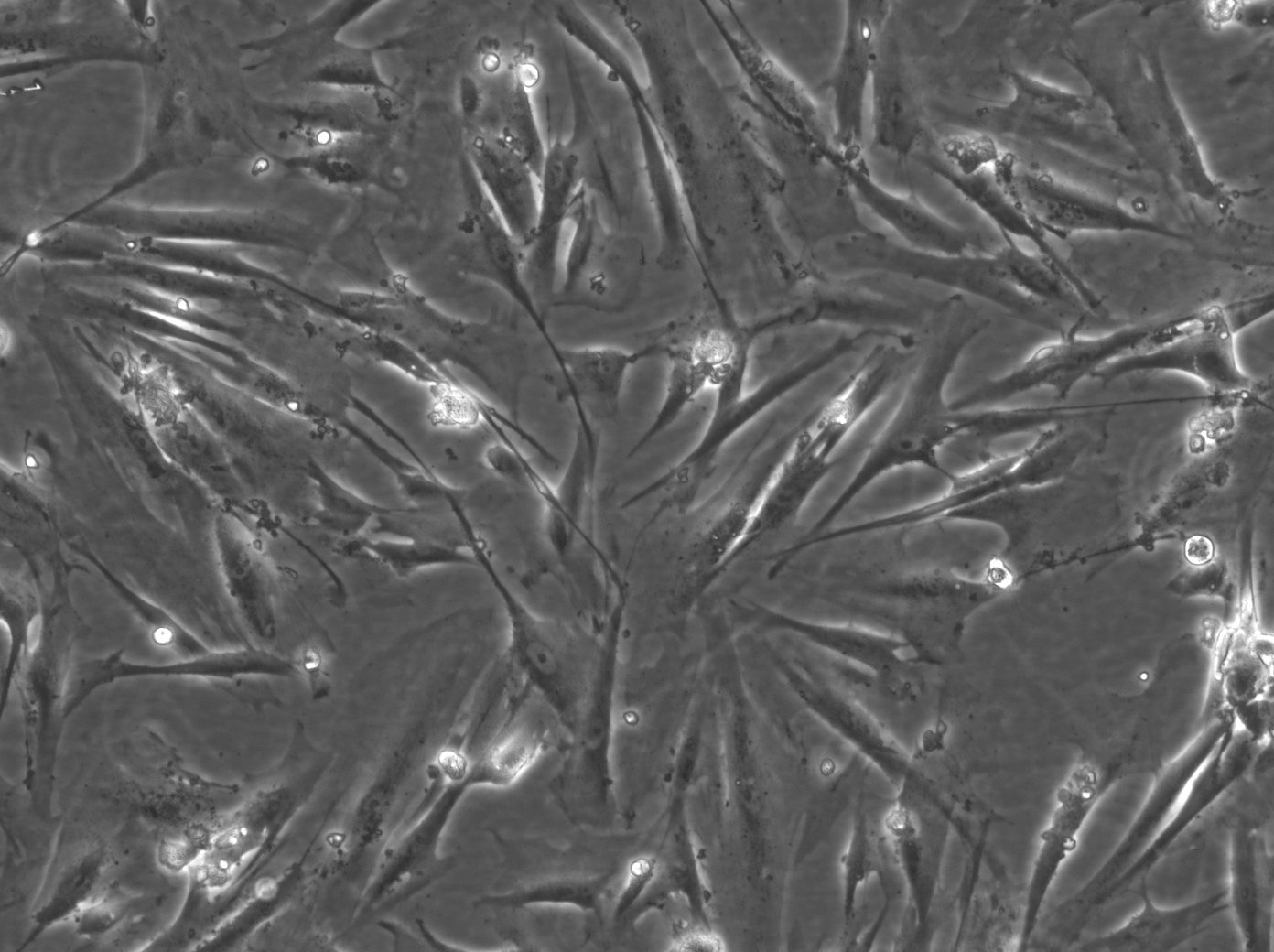
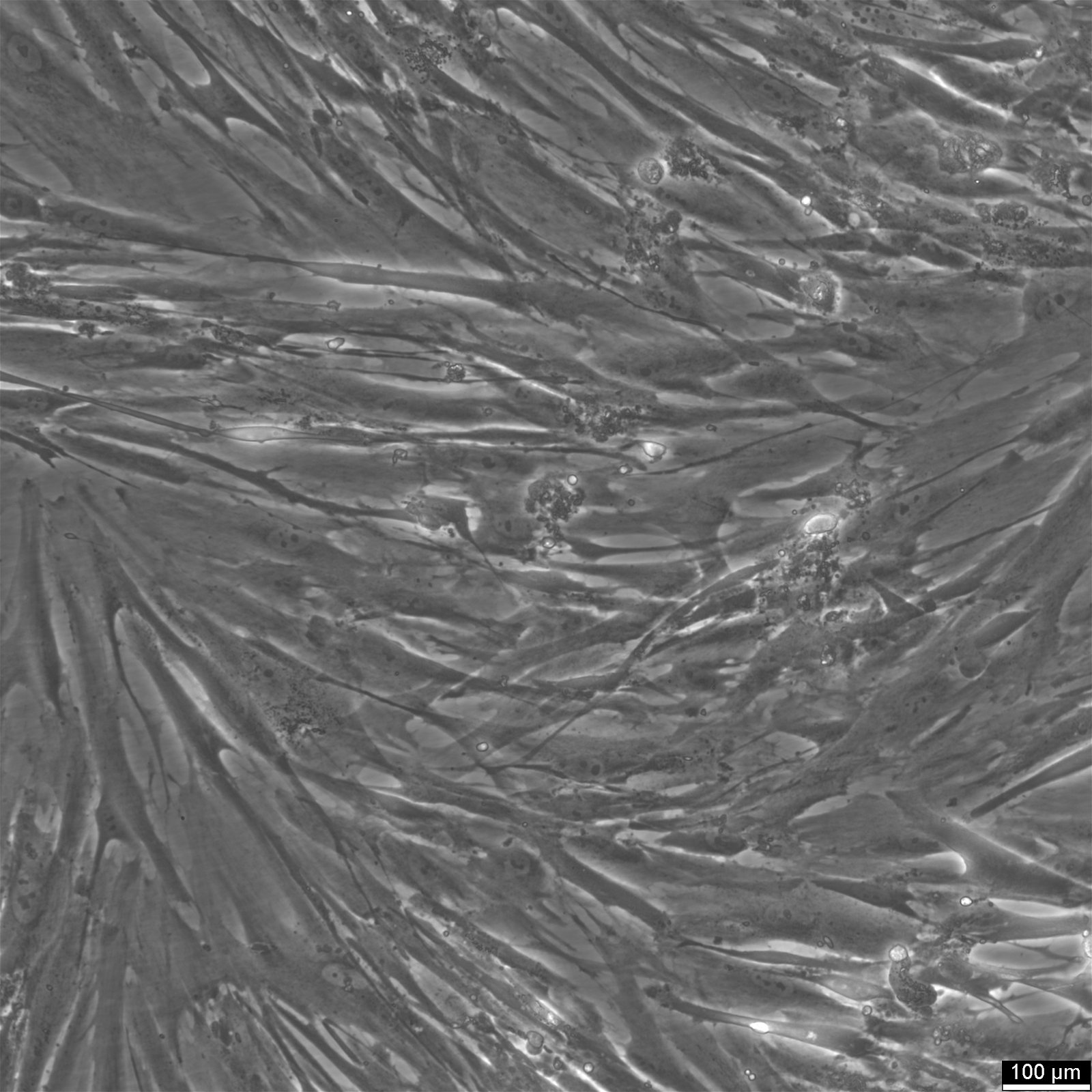












WI-38 cells and their role in vaccine production
| Description | The WI-38 cell line, derived from the fetal lung tissue of a 3-month-old fetus obtained from an elective abortion in Sweden in 1962, represents a landmark in medical science, particularly in vaccine production. WI-38 cells have played a crucial role in the development of vaccines for a wide array of virus-based infectious diseases, including poliomyelitis, measles, mumps, rubella, varicella, herpes zoster, adenovirus, rabies, and Hepatitis A, thereby significantly reducing morbidity associated with these conditions. Notably, WI-38 cells have been utilized in the production of several key vaccines, such as Merck's rubella and Hepatitis A vaccines, Sanofi Pasteur's Imovax rabies vaccine, and the adenovirus vaccine used by the U.S. military, highlighting their essential role in public health. These cells, characterized by their fibroblast cell type and excellent biocompatibility, offer an optimal environment for the culture of viruses and the production of human virus vaccines. As a human diploid cell line with a finite lifespan of about 50 population doublings and a doubling time of roughly 24 hours, WI-38 cells have been used extensively in biological research, including the study of cellular aging, cancer, and genetics. WI-38 cells further have been instrumental in the field of virology, particularly in supporting the cultivation and study of human viruses. These cells provide a conducive environment for growing viruses extracted from clinical specimens, which is essential for the development of vaccines and for advancing our understanding of viral behaviors and genetics. In summary, WI-38 cells, with their extensive applications in vaccine production remain a cornerstone in the field of virology. Their contribution to the development of cell-derived vaccines and the advancement of primary cells in scientific research underscores their invaluable role in enhancing human health worldwide. |
|---|---|
| Organism | Human |
| Tissue | Lung |
| Synonyms | Wi-38, WI38, Wistar Institute-38, AG06814E, AG06814G, AG06814H, AG06814-J, AG06814J, AG06814-M, AG06814-N |
Properties of the WI-38 cell line
| Age | 3 months gestation |
|---|---|
| Gender | Female |
| Ethnicity | Caucasian |
| Morphology | Epithelial-like |
| Cell type | Fibroblast |
| Growth properties | Adherent |
Specifications
| Citation | WI 38 (Cytion catalog number 300428) |
|---|---|
| Biosafety level | 1 |
Genetics
WI-38 cell culture handling procedures
| Culture Medium | EMEM, w: 2 mM L-Glutamine, w: 1.5 g/L NaHCO3, w: EBSS, w: 1 mM Sodium pyruvate, w: NEAA (Cytion article number 820100c) |
|---|---|
| Medium supplements | Supplement the medium with 10% FBS |
| Passaging solution | Accutase |
| Subculturing | Remove the old medium from the adherent cells and wash them with PBS that lacks calcium and magnesium. For T25 flasks, use 3-5 ml of PBS, and for T75 flasks, use 5-10 ml. Then, cover the cells completely with Accutase, using 1-2 ml for T25 flasks and 2.5 ml for T75 flasks. Let the cells incubate at room temperature for 8-10 minutes to detach them. After incubation, gently mix the cells with 10 ml of medium to resuspend them, then centrifuge at 300xg for 3 minutes. Discard the supernatant, resuspend the cells in fresh medium, and transfer them into new flasks that already contain fresh medium. |
| Freeze medium | CM-1 (Cytion catalog number 800100) or CM-ACF (Cytion catalog number 806100) |
| Handling of cryopreserved cultures | WI 38 cells are shipped in a deep-frozen state on dry ice. Upon receipt, confirm that the vial remains frozen. For storage, place the cryovial immediately at temperatures below -150 degrees. If you plan to culture the cells immediately, swiftly thaw the vial by shaking it in a 37 degrees water bath with clean water and an antimicrobial agent for 40-60 seconds. Remove the vial once a small ice clump persists, ensuring it remains cold. Proceed with all subsequent steps under aseptic conditions. In a sterile flow hood, disinfect the cryovial with 70% ethanol. Then, gently open the vial and transfer the cell suspension into a 15 ml centrifuge tube pre-filled with 8 ml of room temperature culture medium. Gently mix the cells. For cell separation, centrifuge at 300 x g for 3 minutes and dispose of the supernatant. Skipping centrifugation is optional, although any residual freezing medium should be removed after 24 hours. Resuspend the pellet gently in 10 ml of fresh culture medium and divide between two T25 culture flasks. Follow the subculture protocol for subsequent steps. |
Quality control
| Sterility | Mycoplasma contamination is rigorously excluded using both PCR-based assays and luminescence-based mycoplasma detection methods. To ensure there is no bacterial, fungal, or yeast contamination, cell cultures are subjected to daily visual inspections. |
|---|---|
| STR profile |
Amelogenin: x,x
|
| HLA alleles |
A*: 02:05:01, 68:01:01
B*: 08:01:01, 58:01:01
C*: 07:01:01, 07:18:01
DRB1*: 11:01:01, 13:02:01
DQA1*: 01:02:01, 05:05:01
DQB1*: 03:01:01, 06:09:01
DPB1*: 03:01:01, 04:01:01
E: 01:01:01, 01:03:01
|
